Sam Brusco, Associate Editor03.28.22
Sky Medical Technology has achieved additional U.S. Food and Drug Administration (FDA) clearance for its new (W3) geko device to increase microcirculatory lower limb soft tissue blood flow in patients with venous sufficiency and/or ischemia.
The new geko device adds a third electrode for better nerve stimulation, comfort, and outcomes. It also increases to two twelve-hour therapeutic doses, whereas the previous (W2) device offered two six-hour doses.
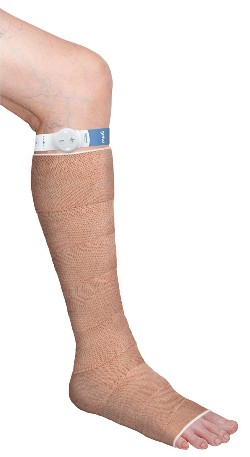
The geko device on the leg.
Recently published studies demonstrated geko generates increased microcirculatory blood flow in lower limbs of patients with venous insufficiency and ischemia. The studies measured increase using laser speckle contrast imaging.
"Achieving this latest 510(k) clearance for the geko device (W3) establishes Sky's position as a leading innovator in medtech dedicated to improving patient outcomes,” Sky Medical CEO and founder Bernard Ross told the press. “Conditions such as venous insufficiency and ischemia are therapy areas sorely in need of innovation, and we are pleased to contribute to improvements in this care pathway that make recovery quicker, simpler and more comfortable."
The geko non-invasive knee-worn therapy device is the size of a watch and stimulates the common peroneal nerve to activate calf and foot muscle pumps to increase blood flow in the calf’s deep veins, which according to the company reaches a rate of 60 percent without the patient needing to move. geko operates without external leg pressure.
The new geko device adds a third electrode for better nerve stimulation, comfort, and outcomes. It also increases to two twelve-hour therapeutic doses, whereas the previous (W2) device offered two six-hour doses.

The geko device on the leg.
"Achieving this latest 510(k) clearance for the geko device (W3) establishes Sky's position as a leading innovator in medtech dedicated to improving patient outcomes,” Sky Medical CEO and founder Bernard Ross told the press. “Conditions such as venous insufficiency and ischemia are therapy areas sorely in need of innovation, and we are pleased to contribute to improvements in this care pathway that make recovery quicker, simpler and more comfortable."
The geko non-invasive knee-worn therapy device is the size of a watch and stimulates the common peroneal nerve to activate calf and foot muscle pumps to increase blood flow in the calf’s deep veins, which according to the company reaches a rate of 60 percent without the patient needing to move. geko operates without external leg pressure.













