Sam Brusco, Associate Editor03.28.22
Implantable eye lens and companion delivery systems firm STAAR Surgical has been granted U.S. Food and Drug Administration (FDA) approval for its EVO/EVO+ Visian Implantable Collamer lens (EVO) to correct myopia and myopia with astigmatism.
"Following FDA approval, prospective patients in the U.S. and their doctors may now consider EVO for achieving visual freedom from the limitations, ongoing maintenance and inconvenience associated with glasses and contact lenses,” Caren Mason, president and CEO of STAAR Surgical told the press. “Over one million EVO lenses have already been implanted by doctors outside the U.S. and 99.4% of EVO patients in a survey have stated they would have the procedure again.1 The EVO lens is additive, provides excellent quality of vision day and night, does not cause dry eye syndrome and is removable by a doctor, if ever so desired.2,3,4 Sales of EVO lenses outside the U.S. increased 51% in 2021 and have more than doubled since 2018, which speaks to the increasing choice by patients and our surgeon partners for EVO as the premium and primary solution for refractive vision correction. Surgeons will be trained and certified for EVO, and begin implanting EVO in the coming days and weeks in select cities. Commercialization of EVO in the U.S. will begin immediately and be supported by a nationwide advertising, marketing and public relations campaign. We wish to express our great appreciation for the outstanding work of our esteemed principal investigator surgeons and their staffs who enrolled patients, implanted and conducted follow-up per the clinical trial protocol during the most challenging of times.”
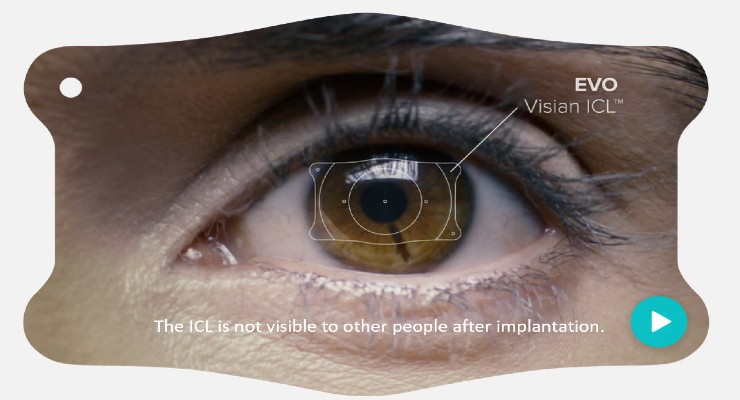
EVOs are inserted in the eye’s posterior chamber behind the iris and in front of the natural crystalline lens. EVO is a lens-based alternative for refractive error correction/reduction for people using glasses or contact lenses for distance vision correction.
A U.S. clinical study confirmed the EVO myopia lens family’s safety and over 100 clinical papers are available assessing the Visian ICL lens family’s safety and efficacy.
Scott D. Barnes, MD, chief medical officer of STAAR Surgical, stated, “We are thrilled to make EVO available to U.S. surgeons and patients seeking a proven and premium option to glasses, contact lenses or laser vision correction. Today’s announcement is especially important because the prevalence of myopia is increasing quickly, and COVID precautions have presented additional challenges to people wearing glasses and/or contact lenses. EVO adds an important tool for the ophthalmic surgeon seeking to help improve a patient’s quality of life. Different from LASIK, the EVO lens is added to the patient’s eye through a relatively quick surgical procedure where there is no removal of corneal tissue. Moreover, the EVO lens is removable by a doctor if ever desired. Results from our recent U.S. clinical trial are consistent with the more than one million EVO lenses that have already been implanted around the world.”
Reference
1 Packer, The Implantable Collamer Lens with a central port: review of the literature, Clinical Ophthalmology, 2018.
2 EVO/EVO+ ICL DFU and PIB.
3 Martinez-Plazs E, Lopez-Miguel A, Lopez-De La Rosa A, et al. Effect of the EVO+ Visian Phakic Implantable Collamer Lens on Visual Performance and Quality of Vision and Life, Am J Ophthalmol 2021;226: 117–125.
4 Naves, J. Carracedo, G. Cacho-Babillo, I. Diadenosine Nucleotid Measurements as Dry-Eye Score in Patients After LASIK and ICL Surgery. Presented at American Society of Cataract and Refractive Surgery (ASCRS) 2012.
"Following FDA approval, prospective patients in the U.S. and their doctors may now consider EVO for achieving visual freedom from the limitations, ongoing maintenance and inconvenience associated with glasses and contact lenses,” Caren Mason, president and CEO of STAAR Surgical told the press. “Over one million EVO lenses have already been implanted by doctors outside the U.S. and 99.4% of EVO patients in a survey have stated they would have the procedure again.1 The EVO lens is additive, provides excellent quality of vision day and night, does not cause dry eye syndrome and is removable by a doctor, if ever so desired.2,3,4 Sales of EVO lenses outside the U.S. increased 51% in 2021 and have more than doubled since 2018, which speaks to the increasing choice by patients and our surgeon partners for EVO as the premium and primary solution for refractive vision correction. Surgeons will be trained and certified for EVO, and begin implanting EVO in the coming days and weeks in select cities. Commercialization of EVO in the U.S. will begin immediately and be supported by a nationwide advertising, marketing and public relations campaign. We wish to express our great appreciation for the outstanding work of our esteemed principal investigator surgeons and their staffs who enrolled patients, implanted and conducted follow-up per the clinical trial protocol during the most challenging of times.”
EVOs are inserted in the eye’s posterior chamber behind the iris and in front of the natural crystalline lens. EVO is a lens-based alternative for refractive error correction/reduction for people using glasses or contact lenses for distance vision correction.
A U.S. clinical study confirmed the EVO myopia lens family’s safety and over 100 clinical papers are available assessing the Visian ICL lens family’s safety and efficacy.
Scott D. Barnes, MD, chief medical officer of STAAR Surgical, stated, “We are thrilled to make EVO available to U.S. surgeons and patients seeking a proven and premium option to glasses, contact lenses or laser vision correction. Today’s announcement is especially important because the prevalence of myopia is increasing quickly, and COVID precautions have presented additional challenges to people wearing glasses and/or contact lenses. EVO adds an important tool for the ophthalmic surgeon seeking to help improve a patient’s quality of life. Different from LASIK, the EVO lens is added to the patient’s eye through a relatively quick surgical procedure where there is no removal of corneal tissue. Moreover, the EVO lens is removable by a doctor if ever desired. Results from our recent U.S. clinical trial are consistent with the more than one million EVO lenses that have already been implanted around the world.”
Reference
1 Packer, The Implantable Collamer Lens with a central port: review of the literature, Clinical Ophthalmology, 2018.
2 EVO/EVO+ ICL DFU and PIB.
3 Martinez-Plazs E, Lopez-Miguel A, Lopez-De La Rosa A, et al. Effect of the EVO+ Visian Phakic Implantable Collamer Lens on Visual Performance and Quality of Vision and Life, Am J Ophthalmol 2021;226: 117–125.
4 Naves, J. Carracedo, G. Cacho-Babillo, I. Diadenosine Nucleotid Measurements as Dry-Eye Score in Patients After LASIK and ICL Surgery. Presented at American Society of Cataract and Refractive Surgery (ASCRS) 2012.