Sam Brusco, Associate Editor03.15.22
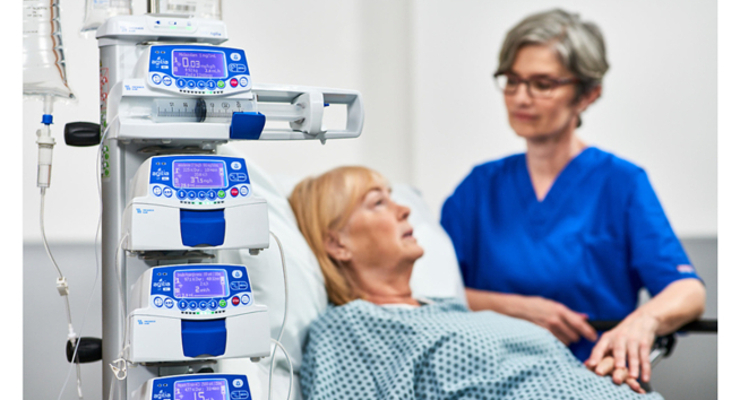
Fresenius Kabi has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its wireless Agilia Connect infusion system, which includes the Agilia volumetric and syringe pumps with Vigilant software suite and Master Med technology.
The Agilia Connect pumps are the first to be cleared by following TIR101 standards, according to the company. The Association for the Advancement of Medical Instrumentation (AAMI) developed the standard last year.
Agilia Connect’s software suite includes drug library software, infusion analytics software, and maintenance/calibration software. The allows centralized distribution of drug libraries, infusion data warehousing for reporting and analysis, and wireless device maintenance and calibration.
“Both patients and caregivers benefit from smarter devices that make healthcare safer and easier,” John Ducker, president and CEO of Fresenius Kabi USA told the press. “As a company with more than 35 years of global infusion technology expertise we look forward to establishing Fresenius Kabi as the partner of choice for infusion therapy in the U.S.”
Agilia features customized master drug libraries based on approved medication protocols. These can be distributed to an entire fleet of Agilia Connect pumps safely and securely. Infusion data can also be configured on demand into reports for independent performance analyses across multiple facilities.
The Agilia Connect pumps are the first to be cleared by following TIR101 standards, according to the company. The Association for the Advancement of Medical Instrumentation (AAMI) developed the standard last year.
Agilia Connect’s software suite includes drug library software, infusion analytics software, and maintenance/calibration software. The allows centralized distribution of drug libraries, infusion data warehousing for reporting and analysis, and wireless device maintenance and calibration.
“Both patients and caregivers benefit from smarter devices that make healthcare safer and easier,” John Ducker, president and CEO of Fresenius Kabi USA told the press. “As a company with more than 35 years of global infusion technology expertise we look forward to establishing Fresenius Kabi as the partner of choice for infusion therapy in the U.S.”
Agilia features customized master drug libraries based on approved medication protocols. These can be distributed to an entire fleet of Agilia Connect pumps safely and securely. Infusion data can also be configured on demand into reports for independent performance analyses across multiple facilities.