Sam Brusco, Associate Editor02.22.22
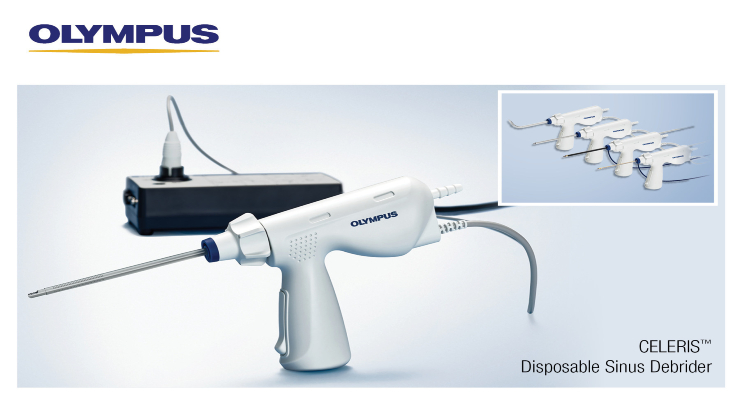
Olympus has received U.S. Food and Drug Administration (FDA) clearance for its CELERIS single-use sinus debrider system. It’s indicated for bone and soft tissue cutting, coagulation, debriding, and removal in general ENT and sinus/rhinology procedures. Specific procedures include turbinoplasty, polypectomy, and endoscopic sinus surgery.
CELERIS’ design allows less invasive procedures in almost any treatment room, and eliminates reprocessing and maintenance costs associated with a full debrider system.
It’s available as a standard 4 mm device, along with 2 mm and 4 mm devices with bipolar capability to manage incidental breeding without needing to change instruments.
A 4 mm device with user-bendable tip can replace multiple blade changes during a normal procedure. It can be bent up to 60 degrees as well.
"While a full debrider system may be the right device in certain cases, the single-use CELERIS debrider offers physicians another option for most of their routine procedures," Dana Currier, VP and Business Unit Leader ENT for Olympus Corp. of the Americas told the press. "Its simple setup and single-use design allows physicians to treat patients in the more comfortable setting of an office treatment room rather than an OR. And it can help improve patient outcomes by offering a bipolar energy option to manage incidental bleeding."
CELERIS’ design allows less invasive procedures in almost any treatment room, and eliminates reprocessing and maintenance costs associated with a full debrider system.
It’s available as a standard 4 mm device, along with 2 mm and 4 mm devices with bipolar capability to manage incidental breeding without needing to change instruments.
A 4 mm device with user-bendable tip can replace multiple blade changes during a normal procedure. It can be bent up to 60 degrees as well.
"While a full debrider system may be the right device in certain cases, the single-use CELERIS debrider offers physicians another option for most of their routine procedures," Dana Currier, VP and Business Unit Leader ENT for Olympus Corp. of the Americas told the press. "Its simple setup and single-use design allows physicians to treat patients in the more comfortable setting of an office treatment room rather than an OR. And it can help improve patient outcomes by offering a bipolar energy option to manage incidental bleeding."