Sam Brusco, Associate Editor02.16.22
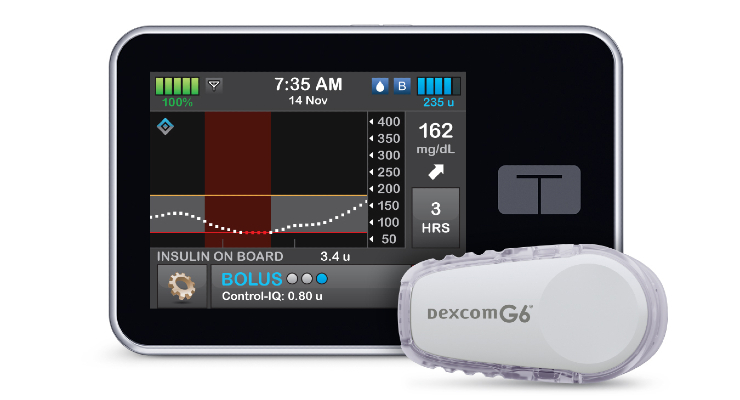
Tandem Diabetes Care today announced U.S. Food and Drug Administration (FDA) clearance for bolus insulin dosing on its t:slim X2 insulin pump via the t:connect mobile app. According to the company, this is the first FDA nod for a smartphone app that can initiate insulin delivery on both iOS and Android systems.
Users can program and cancel bolus insulin requests through a compatible smartphone.
“This FDA clearance further validates our commitment to innovation and the diabetes community by providing one of the most requested feature enhancements,” John Sheridan, president and CEO of Tandem Diabetes Care told the press. “With the improvements in diabetes management provided by Tandem’s Control-IQ technology, giving a meal bolus is now the most common reason a person interacts with their pump, and the ability to do so using a smartphone app offers a convenient and discrete solution.”
The new feature, once released, will be offered via remote software update at no cost to t:slim X2 pump users and in-warranty customers. Tandem Diabetes aims to release the mobile bolus feature throughout the spring in limited groups, then expand the launch later in the summer.
The t:connect mobile app discreetly displays pump info as well as the last 24 hours of glucose trends, pump status changes, and insulin therapy data. It also displays pump alerts and alarms, and securely and wirelessly transmits data to the cloud-based t:connect web app.
The t:slim X2 insulin pump gained new FDA approval for children aged 6-14 in August 2020.
Users can program and cancel bolus insulin requests through a compatible smartphone.
“This FDA clearance further validates our commitment to innovation and the diabetes community by providing one of the most requested feature enhancements,” John Sheridan, president and CEO of Tandem Diabetes Care told the press. “With the improvements in diabetes management provided by Tandem’s Control-IQ technology, giving a meal bolus is now the most common reason a person interacts with their pump, and the ability to do so using a smartphone app offers a convenient and discrete solution.”
The new feature, once released, will be offered via remote software update at no cost to t:slim X2 pump users and in-warranty customers. Tandem Diabetes aims to release the mobile bolus feature throughout the spring in limited groups, then expand the launch later in the summer.
The t:connect mobile app discreetly displays pump info as well as the last 24 hours of glucose trends, pump status changes, and insulin therapy data. It also displays pump alerts and alarms, and securely and wirelessly transmits data to the cloud-based t:connect web app.
The t:slim X2 insulin pump gained new FDA approval for children aged 6-14 in August 2020.