Global Instrumentation LLC01.10.22
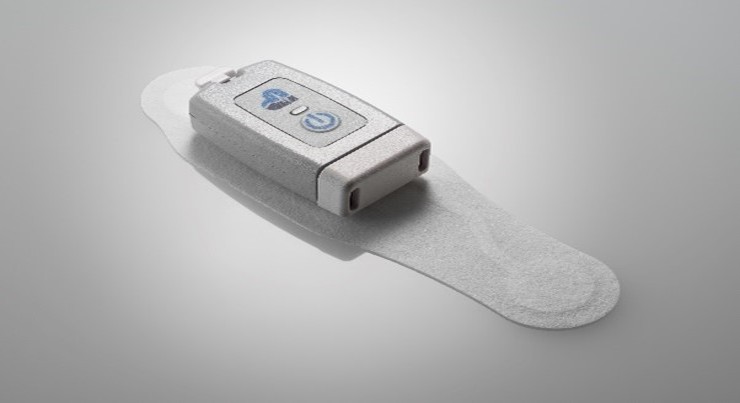
Global Instrumentation LLC has received CE Mark certification for the M5 Wearable ECG Recorder, which affirms that the U.S. manufactured device has met EU safety, health and environmental protection requirements, and can now be marketed and sold in Europe. The M5 device offers research organizations and physicians a reliable, shower-proof, and patient-friendly ECG sensor for precise long-term ECG data collection in the patient’s home and ambulatory environments.
“Receiving the CE marking for the M5 Recorder is a major achievement for Global Instrumentation and a strong endorsement that the performance, quality, and safety of the M5 Recorder meets the essential requirements of all relevant European Medical Device Regulations (MDR)” said Jim DeMaso, Global Instrumentation president.
Fast Facts:
Doug Linquest, vice president of Marketing and Business Development, said, “Our partners in the pharmaceutical research industry support trials that are global in scope. By gaining approval to market the M5 Recorder in Europe, we extend the reach of our cardiac safety platform and ensure that our partners can deploy our full portfolio for clinical trial use in key European geographies.”
The M5 Wearable ECG Recorder received 510(k) clearance from the U.S. Food and Drug Administration in January 2021.
Global Instrumentation LLC provides customized cardiac diagnostic and monitoring solutions to clinical researchers, cardiac monitoring service providers, and medical technology developers seeking precision ECG data collection and flexible data management tools. Through its research, design, and innovation capabilities, the company partners closely with customers to help enable the delivery of better healthcare. Global Instrumentation was founded in 2002 and is based in Manlius, N.Y.
“Receiving the CE marking for the M5 Recorder is a major achievement for Global Instrumentation and a strong endorsement that the performance, quality, and safety of the M5 Recorder meets the essential requirements of all relevant European Medical Device Regulations (MDR)” said Jim DeMaso, Global Instrumentation president.
Fast Facts:
- The M5 Recorder is water-resistant, weighs 26 grams, and can store up to 21 days of three-channel ECG recordings.
- The device is used in tandem with the flagship M12A Enterprise software platform, leveraging the software’s full cardiac data management and analysis capability for clinical-grade remote ECG data collection.
- Unlike many other long-term ambulatory recorders, the M5 can be worn with a patch electrode or small, lightweight patient cables utilizing two to five standard electrodes, providing new levels of flexibility for varying ECG assessment needs.
- It is configurable for sample rate, channels, and duration followed by extensive EGC analysis and report generation capability at the conclusion of the study.
- In any configuration chosen, electrodes can be replaced by the patient during a study with no disruption to data collection or the requirement to return to a facility to extend the recording period.
Doug Linquest, vice president of Marketing and Business Development, said, “Our partners in the pharmaceutical research industry support trials that are global in scope. By gaining approval to market the M5 Recorder in Europe, we extend the reach of our cardiac safety platform and ensure that our partners can deploy our full portfolio for clinical trial use in key European geographies.”
The M5 Wearable ECG Recorder received 510(k) clearance from the U.S. Food and Drug Administration in January 2021.
Global Instrumentation LLC provides customized cardiac diagnostic and monitoring solutions to clinical researchers, cardiac monitoring service providers, and medical technology developers seeking precision ECG data collection and flexible data management tools. Through its research, design, and innovation capabilities, the company partners closely with customers to help enable the delivery of better healthcare. Global Instrumentation was founded in 2002 and is based in Manlius, N.Y.