Boston Scientific06.08.20
Boston Scientific today announced that the Centers for Medicare & Medicaid Services (CMS) has approved its application for a transitional pass-through (TPT) payment category to describe single-use endoscopes, including the Exalt Model D Single-Use Duodenoscope, under the Medicare hospital outpatient prospective payment system (OPPS). The intent of TPT payment is to facilitate Medicare beneficiary access to the advantages of new and innovative devices by allowing for adequate payment for these new devices while the necessary cost data is collected to incorporate the costs for these devices into the procedure Ambulatory Payment Classifications (APC) rate.
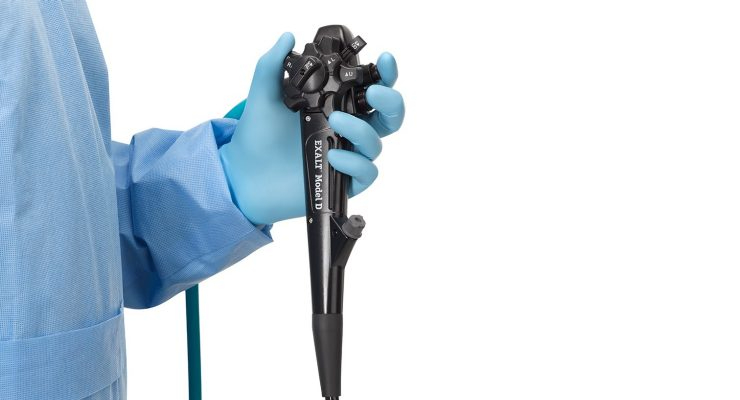
The new device transitional pass-through code (C1748) may be used to bill for EXALT Model D when it is used in treatment of Medicare beneficiaries in the hospital outpatient setting starting July 1, 2020.
The Exalt Model D Single-Use Duodenoscope is a single-use, flexible duodenoscope cleared by the US. Food and Drug Administration (FDA). Duodenoscopes are used in endoscopic retrograde cholangiopancreatography (ERCP) procedures to diagnose and treat various pancreatic and biliary conditions. Every year, more than 700,000 ERCP procedures are performed in the U.S. and approximately 1.5 million are completed worldwide.1
Notably, the FDA has recommended that providers transition to duodenoscopes with disposable components or fully disposable devices, when they are available.2
The FDA granted Exalt Model D its Breakthrough Devices Designation through a program intended to expedite the development and prioritize the review of certain medical devices that provide for more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases or conditions. Breakthrough Device Designation provides patients more timely access to novel medical devices such as Exalt Model D.
Since January 2020, CMS has provided an alternative pathway for innovative technologies that have received FDA marketing authorization and Breakthrough Devices Designation to qualify for device pass-through payment. Exalt Model D is among the first devices to receive TPT approval by CMS via this pathway.
"The approval of this new payment category in the outpatient setting will help ensure healthcare providers have access to Exalt Model D at a time when there is heightened awareness of the need to eliminate infection risk for patients, physicians and hospital staff. With Exalt Model D, physicians can use a new, sterile duodenoscope for every procedure," said Brian Dunkin, M.D., chief medical officer, endoscopy, Boston Scientific. "Boston Scientific has been dedicated to advancing the care of pancreaticobiliary diseases for over 30 years, and we are proud that the Centers for Medicare & Medicaid Services, the country's largest payer for health care, recognizes the importance of our technology to its beneficiaries."
References:
1. Internal Estimate
2. United States Food and Drug Administration website: " The FDA is Recommending Transition to Duodenoscopes with Innovative Designs to Enhance Safety: FDA Safety Communication" April 10, 2020
The new device transitional pass-through code (C1748) may be used to bill for EXALT Model D when it is used in treatment of Medicare beneficiaries in the hospital outpatient setting starting July 1, 2020.
The Exalt Model D Single-Use Duodenoscope is a single-use, flexible duodenoscope cleared by the US. Food and Drug Administration (FDA). Duodenoscopes are used in endoscopic retrograde cholangiopancreatography (ERCP) procedures to diagnose and treat various pancreatic and biliary conditions. Every year, more than 700,000 ERCP procedures are performed in the U.S. and approximately 1.5 million are completed worldwide.1
Notably, the FDA has recommended that providers transition to duodenoscopes with disposable components or fully disposable devices, when they are available.2
The FDA granted Exalt Model D its Breakthrough Devices Designation through a program intended to expedite the development and prioritize the review of certain medical devices that provide for more effective treatment or diagnosis for life-threatening or irreversibly debilitating diseases or conditions. Breakthrough Device Designation provides patients more timely access to novel medical devices such as Exalt Model D.
Since January 2020, CMS has provided an alternative pathway for innovative technologies that have received FDA marketing authorization and Breakthrough Devices Designation to qualify for device pass-through payment. Exalt Model D is among the first devices to receive TPT approval by CMS via this pathway.
"The approval of this new payment category in the outpatient setting will help ensure healthcare providers have access to Exalt Model D at a time when there is heightened awareness of the need to eliminate infection risk for patients, physicians and hospital staff. With Exalt Model D, physicians can use a new, sterile duodenoscope for every procedure," said Brian Dunkin, M.D., chief medical officer, endoscopy, Boston Scientific. "Boston Scientific has been dedicated to advancing the care of pancreaticobiliary diseases for over 30 years, and we are proud that the Centers for Medicare & Medicaid Services, the country's largest payer for health care, recognizes the importance of our technology to its beneficiaries."
References:
1. Internal Estimate
2. United States Food and Drug Administration website: " The FDA is Recommending Transition to Duodenoscopes with Innovative Designs to Enhance Safety: FDA Safety Communication" April 10, 2020