Business Wire05.21.20
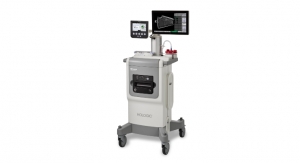
Hologic has announced the 1,000th placement of a Panther system, the company’s automated molecular instrument, in a United States diagnostics laboratory. The 1,000th Panther was unveiled at PathGroup, one of the largest private providers of comprehensive anatomic, clinical and molecular pathology laboratory services in the country. Founded in 1965 and based in Nashville, PathGroup delivers diagnostic serves to more than 15,000 physician clients, hospitals and surgery centers across the United States.
“We are pleased that Hologic has chosen PathGroup as the recipient of the 1,000th Panther molecular diagnostic testing system in the United States,” said Dr. Michael Hanbury, executive vice president and chief operating officer at PathGroup. “This placement is representative of the multi-decade collaboration between our companies to advance awareness, diagnosis and treatment options in women’s health. PathGroup was one of the first U.S. medical laboratories to offer Hologic’s ThinPrep Pap Test and, for decades, has offered clinicians and patients one of the most advanced women’s health test portfolios in the country. PathGroup installed its first Hologic Panther system in 2012, and continues to expand its infectious disease, cytology and molecular pathology services to clients and hospital systems.”
“Our laboratory partners are the unsung heroes of medicine,” said Kevin Thornal, president, Diagnostic Solutions Division at Hologic. “Every day they are working nonstop to supply vital information that drives effective patient care. It is truly an honor to support their mission with our large installed base of Panther systems and broad assay menu.”
This year represents the 10-year anniversary of the Panther system, launched in Europe in 2010 and in the U.S. in 2012. Hologic recently launched Panther Scalable Solutions, which adds new configurable options to the Panther system. Panther Scalable Solutions allows laboratories like PathGroup to choose custom configurations to increase operational capacity and testing volumes at their own pace, while building on the flexibility and streamlined user experience of the Panther system. Worldwide, there are more than 1,700 Panther systems installed for diagnostic use. In addition, more than 500 Procleix Panther systems have been placed by Grifols, for the screening of plasma supply and donated blood.
The Panther molecular diagnostics system is a best-in-class, fully automated, sample-to-result platform with adaptable workflow options and a consolidated testing menu. It combines women’s health, sexually transmitted infections and viral load testing, which can all be done simultaneously. The Panther Fusion system provides an expanded in-vitro diagnostics (IVD) menu, as well as Open Access functionality to run laboratory developed tests. Hologic’s Panther and Panther Fusion systems now offer 16 U.S. Food and Drug Administration-cleared assays and 20 CE-marked tests that detect more than 20 pathogens.
“We are pleased that Hologic has chosen PathGroup as the recipient of the 1,000th Panther molecular diagnostic testing system in the United States,” said Dr. Michael Hanbury, executive vice president and chief operating officer at PathGroup. “This placement is representative of the multi-decade collaboration between our companies to advance awareness, diagnosis and treatment options in women’s health. PathGroup was one of the first U.S. medical laboratories to offer Hologic’s ThinPrep Pap Test and, for decades, has offered clinicians and patients one of the most advanced women’s health test portfolios in the country. PathGroup installed its first Hologic Panther system in 2012, and continues to expand its infectious disease, cytology and molecular pathology services to clients and hospital systems.”
“Our laboratory partners are the unsung heroes of medicine,” said Kevin Thornal, president, Diagnostic Solutions Division at Hologic. “Every day they are working nonstop to supply vital information that drives effective patient care. It is truly an honor to support their mission with our large installed base of Panther systems and broad assay menu.”
This year represents the 10-year anniversary of the Panther system, launched in Europe in 2010 and in the U.S. in 2012. Hologic recently launched Panther Scalable Solutions, which adds new configurable options to the Panther system. Panther Scalable Solutions allows laboratories like PathGroup to choose custom configurations to increase operational capacity and testing volumes at their own pace, while building on the flexibility and streamlined user experience of the Panther system. Worldwide, there are more than 1,700 Panther systems installed for diagnostic use. In addition, more than 500 Procleix Panther systems have been placed by Grifols, for the screening of plasma supply and donated blood.
The Panther molecular diagnostics system is a best-in-class, fully automated, sample-to-result platform with adaptable workflow options and a consolidated testing menu. It combines women’s health, sexually transmitted infections and viral load testing, which can all be done simultaneously. The Panther Fusion system provides an expanded in-vitro diagnostics (IVD) menu, as well as Open Access functionality to run laboratory developed tests. Hologic’s Panther and Panther Fusion systems now offer 16 U.S. Food and Drug Administration-cleared assays and 20 CE-marked tests that detect more than 20 pathogens.