Business Wire08.15.19
In organ donation, time is crucial—both for the person awaiting a life-saving organ, and for the donated organ, which must remain viable long enough to be transported and transplanted. To buy the precious hours needed to save a life, San Antonio, Texas-based Vascular Perfusion Solutions (VPS) has developed ULiSSES, a first-of-its-kind medical device. The device is designed to preserve and resuscitate vascularized tissue— organs and limbs—for more than 24 hours.
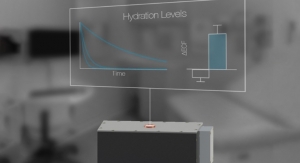
More than 20 people die every day, due to the lack of available organs for transplant, according to the American Transplant Foundation. Per the Organ Donation and Transplantation Alliance, donated organs have an extremely short shelf life. More than 50 percent of all donated organs fail to reach transplant patients in time, and are thus rejected. When it comes to specific organs, the rejection numbers jump significantly, with more than 70 percent of hearts and 80 percent of lungs unable to be transported in time. With the ULiSSES device, a heart can have its viability extended from only four hours, to more than 24 hours. Other organs gain a similar advantage. To achieve this advance, VPS is working to further develop the ULiSSES technology, which is licensed by the University of Texas Health Science Center at San Antonio.
In addition to its organ transport capabilities, VPS is exploring the use of the ULiSSES device to save limbs that are lost in accidents, or on the battlefield. Each year, there are 835,500 accident-related amputations suffered by civilians and combat troops. Until the creation of the ULiSSES device, there has been no major advancement in limb recovery and preservation technology. Additionally, the ULiSSES device can serve as a bioreactor, to quickly grow skin from a recipient’s own cells. Burn victims would benefit immensely from having a supply of their own skin to use to cover damaged areas. VPS plans to use the U.S. Food and Drug Administration’s Expedited Access Program to begin a streamlined review process, to get the device into early clinical use.
“With ULiSSES, the potential impact for patients—more organs for transplant, better compatibility matches, and fewer organ rejections—is tremendous,” said Tom DeBrooke, VPS president and CEO.
VPS has a Cooperative Research and Development Agreement in place with the University of Texas Health Science Center at San Antonio and the U.S. Army’s Institute of Surgical Research (ISR), also in San Antonio. VPS also has a joint operating agreement with the U.S. Army, with animal testing to be conducted by the ISR. In addition to these partnerships, VPS is also working with Harvard Medical School, Massachusetts General Hospital, and the U.S. Department of Defense. In support of additional work VPS, along with other partners, has been the recipient of a Joint Warfighter Medical Research grant from the U.S. Department of Defense.
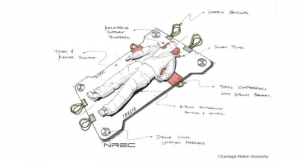
As a device, ULiSSES is highly portable, single-use/disposable, simple to operate, light-weight, and inexpensive. It can easily be transported in a commercial airliner—stored in an overhead compartment, or under the seat. The commercial version of the device will be designed for efficient operation, using only a small oxygen cylinder for power and active perfusion, to allow transport of organs or limbs anywhere in the world. On the battlefield, the ULiSSES device offers a flexible casing option that uses the same fluid carried by medics in common IV bags, to preserve an avulsed hand, arm, or leg until a warfighter can have it reattached at a regular hospital. Without this technology, the limb would be lost, and an artificial substitute would be the only option.
Vascular Perfusion Solutions Inc. is a development and early-stage company formed to commercialize new, unique, and innovative oxygenated perfusion preservation and resuscitation solutions for the body’s limbs, organs, and other vascularized tissue.
More than 20 people die every day, due to the lack of available organs for transplant, according to the American Transplant Foundation. Per the Organ Donation and Transplantation Alliance, donated organs have an extremely short shelf life. More than 50 percent of all donated organs fail to reach transplant patients in time, and are thus rejected. When it comes to specific organs, the rejection numbers jump significantly, with more than 70 percent of hearts and 80 percent of lungs unable to be transported in time. With the ULiSSES device, a heart can have its viability extended from only four hours, to more than 24 hours. Other organs gain a similar advantage. To achieve this advance, VPS is working to further develop the ULiSSES technology, which is licensed by the University of Texas Health Science Center at San Antonio.
In addition to its organ transport capabilities, VPS is exploring the use of the ULiSSES device to save limbs that are lost in accidents, or on the battlefield. Each year, there are 835,500 accident-related amputations suffered by civilians and combat troops. Until the creation of the ULiSSES device, there has been no major advancement in limb recovery and preservation technology. Additionally, the ULiSSES device can serve as a bioreactor, to quickly grow skin from a recipient’s own cells. Burn victims would benefit immensely from having a supply of their own skin to use to cover damaged areas. VPS plans to use the U.S. Food and Drug Administration’s Expedited Access Program to begin a streamlined review process, to get the device into early clinical use.
“With ULiSSES, the potential impact for patients—more organs for transplant, better compatibility matches, and fewer organ rejections—is tremendous,” said Tom DeBrooke, VPS president and CEO.
VPS has a Cooperative Research and Development Agreement in place with the University of Texas Health Science Center at San Antonio and the U.S. Army’s Institute of Surgical Research (ISR), also in San Antonio. VPS also has a joint operating agreement with the U.S. Army, with animal testing to be conducted by the ISR. In addition to these partnerships, VPS is also working with Harvard Medical School, Massachusetts General Hospital, and the U.S. Department of Defense. In support of additional work VPS, along with other partners, has been the recipient of a Joint Warfighter Medical Research grant from the U.S. Department of Defense.
As a device, ULiSSES is highly portable, single-use/disposable, simple to operate, light-weight, and inexpensive. It can easily be transported in a commercial airliner—stored in an overhead compartment, or under the seat. The commercial version of the device will be designed for efficient operation, using only a small oxygen cylinder for power and active perfusion, to allow transport of organs or limbs anywhere in the world. On the battlefield, the ULiSSES device offers a flexible casing option that uses the same fluid carried by medics in common IV bags, to preserve an avulsed hand, arm, or leg until a warfighter can have it reattached at a regular hospital. Without this technology, the limb would be lost, and an artificial substitute would be the only option.
Vascular Perfusion Solutions Inc. is a development and early-stage company formed to commercialize new, unique, and innovative oxygenated perfusion preservation and resuscitation solutions for the body’s limbs, organs, and other vascularized tissue.