Business Wire06.25.18
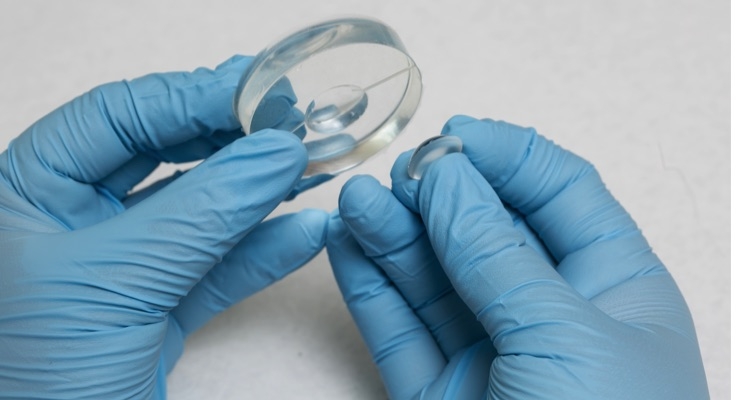
New, drug-eluting soft contact lens technology, has shown to deliver a number of different drugs directly to the eye and may soon improve post-operative recovery for millions of people who undergo cataract and LASIK surgery or suffer corneal abrasions every year.
The new, extended-wear contact, developed by Biomedical researchers at Rowan University and Auburn University, controls the release of anti-inflammatory, anti-biotic and pain reducing drugs that are delivered evenly over time. The new technology can potentially reduce recovery time and post-operative complications, thereby reducing healthcare costs.
“Patients who undergo eye surgery for cataracts, LASIK or who suffer corneal abrasions can experience longer healing times, complications, and even infections if proper care, including administering eye drops, is not properly followed,” said Mark E. Byrne, Ph.D., founding head and professor of Biomedical Engineering at Rowan University, inventor of the technology, and previously a distinguished professor at Auburn University and chief technical officer of OcuMedic Inc. “Our research shows the new contact lens technology is expected to lead to higher efficacy for patients compared to topical eye drops by improving compliance and mitigating concentration peaks and valleys associated with multiple drops.”
“In addition to serving patients who have undergone surgery, as a platform technology, the therapeutic lens has been applied to other diseases of the eye, including dry eye and glaucoma,” said Keith D. Ignotz, president and CEO of OcuMedic, which is commercializing the technology. “The current therapeutic eye drops market is $16 billion annually. A successful drug-eluting, soft contact lens that could potentially capture a large part of the market is expected to garner significant interest from large pharmaceutical companies.”
OcuMedic is expected to begin clinical studies necessary for U.S. Food and Drug Administration (FDA) approval this year, first as an in-vivo study for the FDA to navigate into the Phase 1 – 2 human trials in 2019.
The technology is covered by nine U.S. patents and is expected to be the first product of its kind on the market, once approved.
Approximately 6 million Americans undergo some type of eye surgery every year—4 million cataract and 1 million post-corneal abrasion surgeries, and 800,000 Lasik procedures. All patients require post-operative care, which includes eye drops that can wash out quickly with only about 5 percent of the therapy reaching the target tissue. Eye drops can be hard to administer, and the low delivered concentration of medication tends to aggravate post-operative complications, costing $70 billion per year. Dry eye disease affects greater than 5 million people in the United States and approximately 40 million worldwide, and is the most frequent complaint and reason to visit an eye care professional in the United States. Approximately 3 million Americans suffer from glaucoma; worldwide, more than 60 million people are affected.
Byrne and OcuMedic have received approximately $1.85 million in funding to date, including research funding from the National Institutes of Health. The company recently received an undisclosed amount from a national angel investor group and is staging for a Series A funding in the $4 million range.
OcuMedic Inc. has developed the world’s first drug-eluting therapeutic lens/clear corneal bandage for the delivery of drugs for the global ophthalmic market. The therapeutics lens is a soft contact developed in the most widely used lens material that precisely controls the release of drugs that target post-operative eye care to reduce healing time and complications. The device is a platform technology, with OcuMedic demonstrating controlled release of a number of therapeutics for the duration of wear, targeted at the large and growing surgery, dry eye, and glaucoma markets.
The new, extended-wear contact, developed by Biomedical researchers at Rowan University and Auburn University, controls the release of anti-inflammatory, anti-biotic and pain reducing drugs that are delivered evenly over time. The new technology can potentially reduce recovery time and post-operative complications, thereby reducing healthcare costs.
“Patients who undergo eye surgery for cataracts, LASIK or who suffer corneal abrasions can experience longer healing times, complications, and even infections if proper care, including administering eye drops, is not properly followed,” said Mark E. Byrne, Ph.D., founding head and professor of Biomedical Engineering at Rowan University, inventor of the technology, and previously a distinguished professor at Auburn University and chief technical officer of OcuMedic Inc. “Our research shows the new contact lens technology is expected to lead to higher efficacy for patients compared to topical eye drops by improving compliance and mitigating concentration peaks and valleys associated with multiple drops.”
“In addition to serving patients who have undergone surgery, as a platform technology, the therapeutic lens has been applied to other diseases of the eye, including dry eye and glaucoma,” said Keith D. Ignotz, president and CEO of OcuMedic, which is commercializing the technology. “The current therapeutic eye drops market is $16 billion annually. A successful drug-eluting, soft contact lens that could potentially capture a large part of the market is expected to garner significant interest from large pharmaceutical companies.”
OcuMedic is expected to begin clinical studies necessary for U.S. Food and Drug Administration (FDA) approval this year, first as an in-vivo study for the FDA to navigate into the Phase 1 – 2 human trials in 2019.
The technology is covered by nine U.S. patents and is expected to be the first product of its kind on the market, once approved.
Approximately 6 million Americans undergo some type of eye surgery every year—4 million cataract and 1 million post-corneal abrasion surgeries, and 800,000 Lasik procedures. All patients require post-operative care, which includes eye drops that can wash out quickly with only about 5 percent of the therapy reaching the target tissue. Eye drops can be hard to administer, and the low delivered concentration of medication tends to aggravate post-operative complications, costing $70 billion per year. Dry eye disease affects greater than 5 million people in the United States and approximately 40 million worldwide, and is the most frequent complaint and reason to visit an eye care professional in the United States. Approximately 3 million Americans suffer from glaucoma; worldwide, more than 60 million people are affected.
Byrne and OcuMedic have received approximately $1.85 million in funding to date, including research funding from the National Institutes of Health. The company recently received an undisclosed amount from a national angel investor group and is staging for a Series A funding in the $4 million range.
OcuMedic Inc. has developed the world’s first drug-eluting therapeutic lens/clear corneal bandage for the delivery of drugs for the global ophthalmic market. The therapeutics lens is a soft contact developed in the most widely used lens material that precisely controls the release of drugs that target post-operative eye care to reduce healing time and complications. The device is a platform technology, with OcuMedic demonstrating controlled release of a number of therapeutics for the duration of wear, targeted at the large and growing surgery, dry eye, and glaucoma markets.