Globe Newswire06.18.18
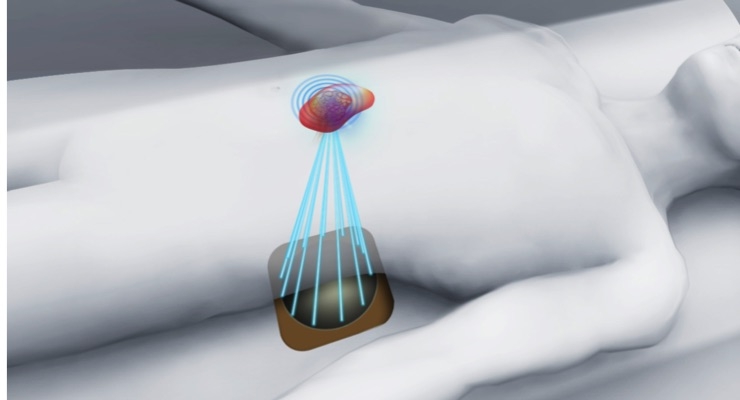
Profound Medical Corp., the only company to provide a therapeutics platform that provides the precision of real-time Magnetic Resonance (MR) imaging combined with the safety and ablation power of directional and focused ultrasound technology for the incision-free ablation of diseased tissue, announced that the Chinese Food and Drug Administration (CFDA) has approved Sonalleve for the non-invasive treatment of uterine fibroids.
Uterine fibroids, or leiomyoma, are the most common non-cancerous tumors of the female reproductive tract in women. Affecting an estimated 20 percent to 50 percent of women over 30 years of age, the disease can cause painful symptoms and abnormal menstrual bleeding. Currently, the most common medical treatment for this disease is hysterectomy. Involving invasive surgery, hospitalization and extensive recovery time, hysterectomy also results in a women’s permanent inability to have children.
Sonalleve is a therapeutic platform that combines real-time MR imaging and thermometry with thermal ultrasound to enable precise and incision-free ablation of diseased tissue. Sonalleve can offer women with uterine fibroids a quick, non-invasive therapy option. Patients usually go home the same day and return to their routines within two days.
“Obtaining CFDA approval for Sonalleve marks a major milestone for our company and provides important additional validation of this powerful, patient-friendly new approach to treating uterine fibroids,” said Arun Menawat, Profound’s CEO. “Our distribution partner, Philips, enjoys a strong brand position in China, having installed MR imaging systems in many of the country’s largest hospitals. That, combined with the recent appointment of Ian Heynen to lead our global sales and marketing function, positions us very well to plan and execute the commercial launch of Sonalleve in this very large and attractive market.”
The Profound Medical Corp. team is committed to creating the powerful combination of real-time MR-guidance as the imaging platform and ultrasound as the energy source for delivering non-invasive ablative tools to clinicians. These key technology pillars, linked with intelligent software and robotics, have the potential to fulfill unmet needs of patients and clinicians in many anatomies and disease states, including prostate cancer, uterine fibroids, and bone metastases.
Profound is commercializing a technology, TULSA-PRO, which combines real-time magnetic resonance imaging with transurethral, robotically-driven therapeutic ultrasound and closed-loop thermal feedback control that is designed to provide precise ablation of the prostate while simultaneously protecting critical surrounding anatomy from potential side effects. TULSA-PRO is CE marked and Profound is currently conducting a pilot commercial launch of the technology in key European and other CE mark jurisdictions. The company is also sponsoring a multicenter, prospective U.S. Food and Drug Administration (FDA)-registered clinical trial, TACT, which, if successful, is expected to support its application to the FDA for clearance to market TULSA-PRO in the United States.
Profound Medical is also commercializing Sonalleve, a CE marked therapeutic platform for the treatment of uterine fibroids and palliative pain treatment of bone metastases. The company is also in the early stages of exploring additional potential treatment markets for Sonalleve, such as non-invasive ablation of abdominal cancers and hyperthermia for cancer therapy, where the technology has been shown to have clinical application.
Uterine fibroids, or leiomyoma, are the most common non-cancerous tumors of the female reproductive tract in women. Affecting an estimated 20 percent to 50 percent of women over 30 years of age, the disease can cause painful symptoms and abnormal menstrual bleeding. Currently, the most common medical treatment for this disease is hysterectomy. Involving invasive surgery, hospitalization and extensive recovery time, hysterectomy also results in a women’s permanent inability to have children.
Sonalleve is a therapeutic platform that combines real-time MR imaging and thermometry with thermal ultrasound to enable precise and incision-free ablation of diseased tissue. Sonalleve can offer women with uterine fibroids a quick, non-invasive therapy option. Patients usually go home the same day and return to their routines within two days.
“Obtaining CFDA approval for Sonalleve marks a major milestone for our company and provides important additional validation of this powerful, patient-friendly new approach to treating uterine fibroids,” said Arun Menawat, Profound’s CEO. “Our distribution partner, Philips, enjoys a strong brand position in China, having installed MR imaging systems in many of the country’s largest hospitals. That, combined with the recent appointment of Ian Heynen to lead our global sales and marketing function, positions us very well to plan and execute the commercial launch of Sonalleve in this very large and attractive market.”
The Profound Medical Corp. team is committed to creating the powerful combination of real-time MR-guidance as the imaging platform and ultrasound as the energy source for delivering non-invasive ablative tools to clinicians. These key technology pillars, linked with intelligent software and robotics, have the potential to fulfill unmet needs of patients and clinicians in many anatomies and disease states, including prostate cancer, uterine fibroids, and bone metastases.
Profound is commercializing a technology, TULSA-PRO, which combines real-time magnetic resonance imaging with transurethral, robotically-driven therapeutic ultrasound and closed-loop thermal feedback control that is designed to provide precise ablation of the prostate while simultaneously protecting critical surrounding anatomy from potential side effects. TULSA-PRO is CE marked and Profound is currently conducting a pilot commercial launch of the technology in key European and other CE mark jurisdictions. The company is also sponsoring a multicenter, prospective U.S. Food and Drug Administration (FDA)-registered clinical trial, TACT, which, if successful, is expected to support its application to the FDA for clearance to market TULSA-PRO in the United States.
Profound Medical is also commercializing Sonalleve, a CE marked therapeutic platform for the treatment of uterine fibroids and palliative pain treatment of bone metastases. The company is also in the early stages of exploring additional potential treatment markets for Sonalleve, such as non-invasive ablation of abdominal cancers and hyperthermia for cancer therapy, where the technology has been shown to have clinical application.