Globe Newswire04.26.18
Akers Biosciences Inc., a developer of rapid health information technologies, has entered into a three-year National Distribution Agreement with Diagnostica Stago Inc. for the sale of the company’s flagship rapid test for Heparin-induced thrombocytopenia (HIT) across the United States.
Stago is a global leader in hemostasis, with more than 20,000 instruments in use and an extensive U.S.-based team dedicated to the sale and support of hemostasis products and equipment to hospitals across the country. Under the agreement, Stago will be marketing and selling Akers Bio’s PIFA PLUSS PF4 Rapid Assay, a single-use test for the detection of Platelet Factor 4 antibodies using a whole blood sample, to its broad customer base of hospital laboratories.
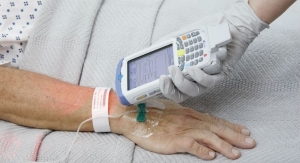
PIFA PLUSS PF4 is designed to determine in under one hour, at (or near) the point-of-care, if a patient being treated with the widely used blood thinner, heparin, may be developing a reaction known as HIT. This clinical syndrome reverses heparin's intended therapeutic effect and transforms it into a clotting agent. Patients suffering HIT (a low platelet count) are at risk of developing limb- and life-threatening complications, so the timely test result provided by the company's device is paramount to effective and cost-efficient clinical decision making. There are approximately 4 million tests for HIT conducted annually in the United States using slower, traditional methods. Akers Bio is aiming to replace a significant portion of these tests with its rapid alternative.
“The addition of Stago to our distributor network is a significant milestone in the commercialization of PIFA PLUSS PF4," said John J. Gormally, CEO of Akers Bio. "Stago is a recognized leader in the field of hemostasis and has a very large and established customer base to which to market our product. Through Stago, Akers Bio will gain access to a specialized sales team who are focused exclusively on buyers of hemostasis-related products. Akers Bio is excited about the potential for near-term U.S. sales growth through this new, additional and specialized channel to market.”
“Stago is pleased to have selected PIFA PLUSS PF4 Rapid Assay as its only rapid test for heparin Platelet Factor 4 antibodies in the U.S.," said Philippe Barroux, North America CEO of Stago. "Our sales team is looking forward to communicating the compelling medical and economic benefits of this uniquely efficient and accurate test for heparin-induced thrombocytopenia to Stago’s customers in line with our commitment to providing market leading hemostasis solutions to improve patient care.”
Akers Bio develops, manufactures, and supplies rapid screening and testing products designed to deliver quicker and more cost-effective healthcare information to healthcare providers and consumers. The company has advanced the science of diagnostics while responding to major shifts in healthcare through the development of several proprietary platform technologies. The company's rapid diagnostic assays can be performed virtually anywhere in minutes when time is of the essence. The company has aligned with major healthcare companies and high-volume medical product distributors to maximize product offerings, and to be a major worldwide competitor in diagnostics.
Stago is a global leader in hemostasis, with more than 20,000 instruments in use and an extensive U.S.-based team dedicated to the sale and support of hemostasis products and equipment to hospitals across the country. Under the agreement, Stago will be marketing and selling Akers Bio’s PIFA PLUSS PF4 Rapid Assay, a single-use test for the detection of Platelet Factor 4 antibodies using a whole blood sample, to its broad customer base of hospital laboratories.
PIFA PLUSS PF4 is designed to determine in under one hour, at (or near) the point-of-care, if a patient being treated with the widely used blood thinner, heparin, may be developing a reaction known as HIT. This clinical syndrome reverses heparin's intended therapeutic effect and transforms it into a clotting agent. Patients suffering HIT (a low platelet count) are at risk of developing limb- and life-threatening complications, so the timely test result provided by the company's device is paramount to effective and cost-efficient clinical decision making. There are approximately 4 million tests for HIT conducted annually in the United States using slower, traditional methods. Akers Bio is aiming to replace a significant portion of these tests with its rapid alternative.
“The addition of Stago to our distributor network is a significant milestone in the commercialization of PIFA PLUSS PF4," said John J. Gormally, CEO of Akers Bio. "Stago is a recognized leader in the field of hemostasis and has a very large and established customer base to which to market our product. Through Stago, Akers Bio will gain access to a specialized sales team who are focused exclusively on buyers of hemostasis-related products. Akers Bio is excited about the potential for near-term U.S. sales growth through this new, additional and specialized channel to market.”
“Stago is pleased to have selected PIFA PLUSS PF4 Rapid Assay as its only rapid test for heparin Platelet Factor 4 antibodies in the U.S.," said Philippe Barroux, North America CEO of Stago. "Our sales team is looking forward to communicating the compelling medical and economic benefits of this uniquely efficient and accurate test for heparin-induced thrombocytopenia to Stago’s customers in line with our commitment to providing market leading hemostasis solutions to improve patient care.”
Akers Bio develops, manufactures, and supplies rapid screening and testing products designed to deliver quicker and more cost-effective healthcare information to healthcare providers and consumers. The company has advanced the science of diagnostics while responding to major shifts in healthcare through the development of several proprietary platform technologies. The company's rapid diagnostic assays can be performed virtually anywhere in minutes when time is of the essence. The company has aligned with major healthcare companies and high-volume medical product distributors to maximize product offerings, and to be a major worldwide competitor in diagnostics.