Business Wire04.20.18
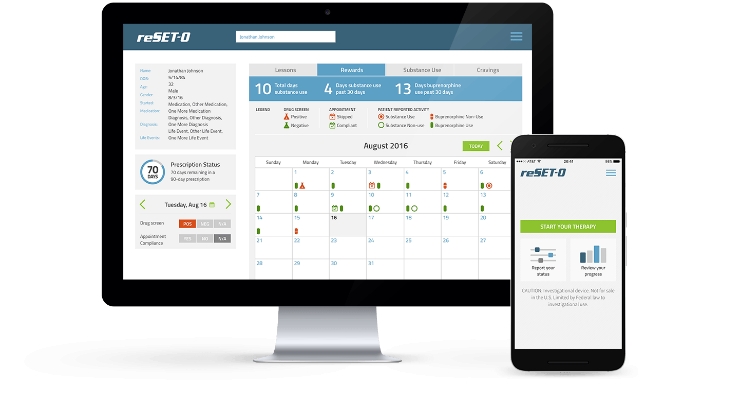
Pear Therapeutics Inc. announced a deal with Sandoz, a division of Novartis, to commercialize its two lead products, reSET and reSET-O. In addition to an upfront payment, the deal includes research and development funding, commercial milestones, and a profit split on net sales of both therapeutics.
reSET was cleared by the FDA in September 2017 for the treatment of patients with Substance Use Disorder, making it the first prescription digital therapeutic cleared with claims to improve clinical outcomes in a disease. reSET-O, a potential prescription digital therapeutic for treating Opioid Use Disorder, was granted Expedited Access Pathway designation in October 2017 and is currently under review by the FDA.
“After a competitive evaluation process among potential partners, Sandoz emerged as the best fit to commercialize reSET and reSET-O,” said Corey McCann, M.D., Ph.D., president and CEO of Pear Therapeutics. “Sandoz has a demonstrated record of successful commercial execution both in the U.S. and globally, has extensive expertise working with clinicians and patients, and will provide a dedicated sales force to market both reSET and reSET-O. We believe they are well-positioned to ensure that these innovative products are available to patients in dire need of more effective treatment options.”
Sandoz will assume responsibility for the global commercial launch of reSET, and reSET-O, including ensuring market access, reimbursement from payors, and providing a dedicated sales force. Pear will continue to develop both digital therapeutics and will also support patient services through its digital hub service.
reSET was cleared by the FDA in September 2017 for the treatment of patients with Substance Use Disorder, making it the first prescription digital therapeutic cleared with claims to improve clinical outcomes in a disease. reSET-O, a potential prescription digital therapeutic for treating Opioid Use Disorder, was granted Expedited Access Pathway designation in October 2017 and is currently under review by the FDA.
“After a competitive evaluation process among potential partners, Sandoz emerged as the best fit to commercialize reSET and reSET-O,” said Corey McCann, M.D., Ph.D., president and CEO of Pear Therapeutics. “Sandoz has a demonstrated record of successful commercial execution both in the U.S. and globally, has extensive expertise working with clinicians and patients, and will provide a dedicated sales force to market both reSET and reSET-O. We believe they are well-positioned to ensure that these innovative products are available to patients in dire need of more effective treatment options.”
Sandoz will assume responsibility for the global commercial launch of reSET, and reSET-O, including ensuring market access, reimbursement from payors, and providing a dedicated sales force. Pear will continue to develop both digital therapeutics and will also support patient services through its digital hub service.