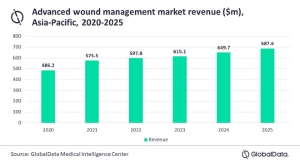
Globe Newswire03.20.18
InspireMD Inc., a developer of embolic prevention systems (EPS)/thrombus management technologies and neurovascular devices, has signed an exclusive distribution, sales, and marketing agreement with Solvit Medical Inc., a South Korean distributor of medical devices and pharmaceuticals. The agreement grants Solvit the exclusive right to market and sell InspireMD’s CGuard Embolic Protection System (EPS) for the treatment of carotid disease upon the Korean Ministry of Food and Drug Safety’s (MFDS) approval.
Under terms of the agreement, Solvit will fund and manage the registration and reimbursement submissions for CGuard EPS in South Korea. All regulatory filings will be in the name of InspireMD Ltd. In addition, Solvit will be contractually committed to purchase a minimum number of CGuard EPS systems annually during the term of the agreement, starting when commercial sales begin in South Korea, which is expected to be in the second quarter of next year. The term of the agreement extends for five years following regulatory approval in South Korea.
Agustin Gago, executive vice president and chief commercial officer of InspireMD, commented, "This agreement is an important step in bringing CGuard EPS to South Korea which represents one of the larger Asian market opportunities, where carotid disease is a major health issue. We believe this agreement, which includes a significant upfront investment on Solvit Medical’s part, is a significant validation of the growing interest in CGuard EPS and our MicroNet technology, both in Asia and globally. We continue to pursue additional strategic partnerships to address other key Asian markets, given the meaningful opportunity in this part of the world.”
Solvit President and CEO K.S. Kim stated, "Becoming the exclusive distributor of CGuard EPS in South Korea is aligned with our business goals, including bringing a best-in-class solution to our physician customers who treat carotid artery disease. This partnership allows us to bring carotid technology to the South Korean medical community, as well as expand our current product offerings to key customers in the interventional, neurovascular and vascular surgery segments, which span all the major clinical specialties that treat carotid artery disease in South Korea. We further view this as the first step in building a strong partnership with InspireMD, as their platform technology is adapted to treat other important vascular disease indications."
InspireMD seeks to utilize its proprietary MicroNet technology to make its products the industry standard for embolic protection and to provide a superior solution to the key clinical issues of current stenting in patients with a high risk of distal embolization, no reflow and major adverse cardiac events. InspireMD intends to pursue applications of this MicroNet technology in coronary, carotid (CGuard), neurovascular, and peripheral artery procedures.
Under terms of the agreement, Solvit will fund and manage the registration and reimbursement submissions for CGuard EPS in South Korea. All regulatory filings will be in the name of InspireMD Ltd. In addition, Solvit will be contractually committed to purchase a minimum number of CGuard EPS systems annually during the term of the agreement, starting when commercial sales begin in South Korea, which is expected to be in the second quarter of next year. The term of the agreement extends for five years following regulatory approval in South Korea.
Agustin Gago, executive vice president and chief commercial officer of InspireMD, commented, "This agreement is an important step in bringing CGuard EPS to South Korea which represents one of the larger Asian market opportunities, where carotid disease is a major health issue. We believe this agreement, which includes a significant upfront investment on Solvit Medical’s part, is a significant validation of the growing interest in CGuard EPS and our MicroNet technology, both in Asia and globally. We continue to pursue additional strategic partnerships to address other key Asian markets, given the meaningful opportunity in this part of the world.”
Solvit President and CEO K.S. Kim stated, "Becoming the exclusive distributor of CGuard EPS in South Korea is aligned with our business goals, including bringing a best-in-class solution to our physician customers who treat carotid artery disease. This partnership allows us to bring carotid technology to the South Korean medical community, as well as expand our current product offerings to key customers in the interventional, neurovascular and vascular surgery segments, which span all the major clinical specialties that treat carotid artery disease in South Korea. We further view this as the first step in building a strong partnership with InspireMD, as their platform technology is adapted to treat other important vascular disease indications."
InspireMD seeks to utilize its proprietary MicroNet technology to make its products the industry standard for embolic protection and to provide a superior solution to the key clinical issues of current stenting in patients with a high risk of distal embolization, no reflow and major adverse cardiac events. InspireMD intends to pursue applications of this MicroNet technology in coronary, carotid (CGuard), neurovascular, and peripheral artery procedures.