Johnson & Johnson03.16.18
Johnson & Johnson Medical Devices Companies* announced today that Biosense Webster Inc., a developer of heart arrhythmia diagnosis and treatment technologies, enrolled and treated the first patient in its SHINE** clinical study in Europe. The multicenter study will evaluate the company’s next generation multi-electrode radiofrequency (RF) balloon catheter for its ability to isolate pulmonary veins when treating paroxysmal or intermittent atrial fibrillation (AF).
The first patient was treated last week at Barts Hospital in London, United Kingdom, by Richard Schilling, M.D., professor of Cardiology. The clinical study will enroll up to 230 patients across multiple sites in Europe.
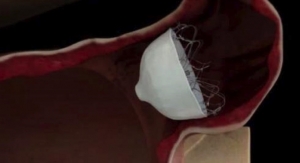
Cardiac ablation is a minimally-invasive procedure that neutralizes parts of the abnormal electrical pathways causing arrhythmia. In the procedure, doctors thread a flexible balloon catheter through blood vessels to the left atrium of the heart. Once there, the balloon is inflated near the pulmonary veins and RF energy is applied to create lines of scar tissue to prevent the pulmonary veins from emitting faulty electrical impulses that can trigger AF.
“This new device may be an important advance in cardiac ablation technology,” said Schilling, who also is president of the British Heart Rhythm Society. “It has unique design characteristics simplifying pulmonary vein isolation that may translate into improved clinical outcomes and procedural efficiencies.”
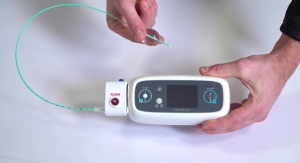
The new RF balloon catheter used in the study is designed to deliver directionally-tailored energy using multiple electrodes. The device will be used in combination with the company’s integrated multi-electrode circular diagnostic catheter to potentially improve procedural efficiency.
“AF is fast becoming one of the world’s most significant public health issues affecting millions of lives and placing a critical burden on healthcare systems,” said Shlomi Nachman, company group chair of Johnson & Johnson Medical Devices Cardiovascular & Specialty Solutions. “Biosense Webster is committed to delivering solutions that help clinicians reach more patients and we look forward to completing this important study and bringing new technologies to the market.”
The Johnson & Johnson Medical Devices Companies* has substantial breadth and depth in surgical and orthopedic technologies and interventional solutions. The company is headquartered in New Brunswick, N.J.
Biosense Webster Inc. develops technology to diagnose and treat heart rhythm disorders. The company is part of the Johnson & Johnson Family of Companies.
*Comprising the surgery, orthopedics, and cardiovascular businesses within Johnson & Johnson’s Medical Devices segment.
**SHINE: Multi-electrode Radiofrequency Balloon Catheter use for the Isolation of the Pulmonary Veins. The device is approved for investigational use only. It is not approved or available for sale.
The first patient was treated last week at Barts Hospital in London, United Kingdom, by Richard Schilling, M.D., professor of Cardiology. The clinical study will enroll up to 230 patients across multiple sites in Europe.
Cardiac ablation is a minimally-invasive procedure that neutralizes parts of the abnormal electrical pathways causing arrhythmia. In the procedure, doctors thread a flexible balloon catheter through blood vessels to the left atrium of the heart. Once there, the balloon is inflated near the pulmonary veins and RF energy is applied to create lines of scar tissue to prevent the pulmonary veins from emitting faulty electrical impulses that can trigger AF.
“This new device may be an important advance in cardiac ablation technology,” said Schilling, who also is president of the British Heart Rhythm Society. “It has unique design characteristics simplifying pulmonary vein isolation that may translate into improved clinical outcomes and procedural efficiencies.”
The new RF balloon catheter used in the study is designed to deliver directionally-tailored energy using multiple electrodes. The device will be used in combination with the company’s integrated multi-electrode circular diagnostic catheter to potentially improve procedural efficiency.
“AF is fast becoming one of the world’s most significant public health issues affecting millions of lives and placing a critical burden on healthcare systems,” said Shlomi Nachman, company group chair of Johnson & Johnson Medical Devices Cardiovascular & Specialty Solutions. “Biosense Webster is committed to delivering solutions that help clinicians reach more patients and we look forward to completing this important study and bringing new technologies to the market.”
The Johnson & Johnson Medical Devices Companies* has substantial breadth and depth in surgical and orthopedic technologies and interventional solutions. The company is headquartered in New Brunswick, N.J.
Biosense Webster Inc. develops technology to diagnose and treat heart rhythm disorders. The company is part of the Johnson & Johnson Family of Companies.
*Comprising the surgery, orthopedics, and cardiovascular businesses within Johnson & Johnson’s Medical Devices segment.
**SHINE: Multi-electrode Radiofrequency Balloon Catheter use for the Isolation of the Pulmonary Veins. The device is approved for investigational use only. It is not approved or available for sale.