Business Wire01.25.18
Corindus Vascular Robotics Inc., a developer of precision vascular robotics, is working with Mayo Clinic in a preclinical study about the use of telestenting. Telestenting, or the remote robotic treatment for percutaneous coronary intervention (PCI), may enable physicians to conduct procedures from virtually any location, opening opportunities for more patients globally to receive the benefits of this lifesaving procedure. The global shortage of PCI-capable operators is significant and continues to be a growing problem.
Mackram F. Eleid, M.D., interventional cardiologist at the Mayo Clinic Department of Cardiovascular Medicine, and associate professor of medicine at the Mayo Clinic College of Medicine, will serve as the primary investigator for the multi-phase study.
Mayo Clinic received a $3.3 million grant from The Leona M. and Harry B. Helmsley Charitable Trust to support the first step of a multi-phase, multi-year development program. Mayo Clinic is working with Corindus to explore telestenting as a solution to the geographic and workforce barriers that exist to provide needed PCI therapy to rural and underserved populations across the globe. Studies will help determine if robotic-assisted PCI can be performed safely and effectively using an off-site remote-controlled system. The CorPath GRX System is currently cleared for robotic-assisted PCI in the cardiac cath lab.
Mark Toland, president and CEO of Corindus, stated, "We are delighted to work on critical research for remote robotics with Mayo Clinic. While PCI is the initial focus for this development program, our long-term goal is to extend this capability to the remote treatment of endovascular disease and stroke. Corindus is committed to developing a high tech cardiovascular model that improves efficiency, integrates the latest technology, and ultimately improves patient care. Telestenting is at the core of this strategy."
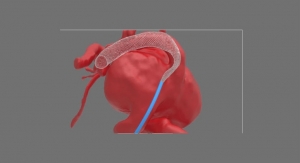
Corindus Vascular Robotics Inc. is a global developer of robotic-assisted vascular intervention technology. The company's CorPath System is the first U.S. Food and Drug Administration-cleared medical device to bring robotic precision to percutaneous coronary interventions. During the procedure, the interventional cardiologist sits at a radiation-shielded workstation to advance guide catheters, stents, and guidewires with millimeter-by-millimeter precision. The workstation allows the physician greater control and the freedom from wearing heavy lead protective equipment that causes musculoskeletal injuries. CorPath GRX is the second generation robotic-assisted PCI technology offering enhancements to the platform by adding important key upgrades that increase precision, improve workflow, and extend the capabilities and range of procedures that can be performed robotically. With the CorPath System, Corindus Vascular Robotics brings robotic precision to interventional procedures to help optimize clinical outcomes and minimize the costs associated with complications of improper stent placement during manual procedures.
Mackram F. Eleid, M.D., interventional cardiologist at the Mayo Clinic Department of Cardiovascular Medicine, and associate professor of medicine at the Mayo Clinic College of Medicine, will serve as the primary investigator for the multi-phase study.
Mayo Clinic received a $3.3 million grant from The Leona M. and Harry B. Helmsley Charitable Trust to support the first step of a multi-phase, multi-year development program. Mayo Clinic is working with Corindus to explore telestenting as a solution to the geographic and workforce barriers that exist to provide needed PCI therapy to rural and underserved populations across the globe. Studies will help determine if robotic-assisted PCI can be performed safely and effectively using an off-site remote-controlled system. The CorPath GRX System is currently cleared for robotic-assisted PCI in the cardiac cath lab.
Mark Toland, president and CEO of Corindus, stated, "We are delighted to work on critical research for remote robotics with Mayo Clinic. While PCI is the initial focus for this development program, our long-term goal is to extend this capability to the remote treatment of endovascular disease and stroke. Corindus is committed to developing a high tech cardiovascular model that improves efficiency, integrates the latest technology, and ultimately improves patient care. Telestenting is at the core of this strategy."
Corindus Vascular Robotics Inc. is a global developer of robotic-assisted vascular intervention technology. The company's CorPath System is the first U.S. Food and Drug Administration-cleared medical device to bring robotic precision to percutaneous coronary interventions. During the procedure, the interventional cardiologist sits at a radiation-shielded workstation to advance guide catheters, stents, and guidewires with millimeter-by-millimeter precision. The workstation allows the physician greater control and the freedom from wearing heavy lead protective equipment that causes musculoskeletal injuries. CorPath GRX is the second generation robotic-assisted PCI technology offering enhancements to the platform by adding important key upgrades that increase precision, improve workflow, and extend the capabilities and range of procedures that can be performed robotically. With the CorPath System, Corindus Vascular Robotics brings robotic precision to interventional procedures to help optimize clinical outcomes and minimize the costs associated with complications of improper stent placement during manual procedures.