BIOCORP01.22.18
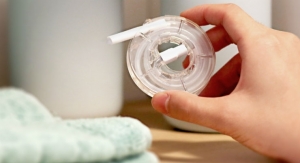
BIOCORP, a French company specializing in the development and manufacturing of medical devices and smart-drug delivery systems, has received CE marking for its smart cap Easylog.
"CE marking for Easylog is a significant step for BIOCORP. Our ambition is to develop and market innovative devices boosting compliance, a key factor for the successful treatment of chronic conditions such as diabetes. Easylog device, which generates interest of many industrial players internationally, brings an extra dimension to the customization of treatments. We are particularly pleased to reach this milestone that confirms our position in the digitalization of drug delivery devices and will contribute to achieve our growth objectives," said Eric Dessertenne, chief operating officer of BIOCORP.
Easylog is a smart sensor that turns all classic pen injectors, disposable and reusable, into connected devices. Easylog captures key treatment information (dose, date, time) and transfers it to a monitoring mobile app. This solution goes hand in hand with a dedicated application that aims to facilitate the life of patients monitoring their treatment.
CE marking has been received for a version compatible with insulin delivery devices. Market launch is planned this through partnerships.
Founded in 2004 in Issoire (near Clermont-Ferrand), France, BIOCORP specializes in developing and manufacturing medical devices and drug delivery systems. With over twenty years of experience and more than 30 manufactured products, BIOCORP is a key player in the industry, providing drug delivery solutions that meet the evolving needs of patients. Today, BIOCORP continues to innovate in medical plastics, its core business, and to market traditional devices (alternative to aluminum capsules, syringe and vial administration systems) that have been an important source of recurring income. Its Internet-connected products include the DataPen, a reusable smart injection pen that automatically transmits data to a mobile app, helping patients to manage their treatment; and treatment management add-ons, which adapt to existing delivery devices and are compatible with most injection pens in the market. The company has a team of 48 employees.
"CE marking for Easylog is a significant step for BIOCORP. Our ambition is to develop and market innovative devices boosting compliance, a key factor for the successful treatment of chronic conditions such as diabetes. Easylog device, which generates interest of many industrial players internationally, brings an extra dimension to the customization of treatments. We are particularly pleased to reach this milestone that confirms our position in the digitalization of drug delivery devices and will contribute to achieve our growth objectives," said Eric Dessertenne, chief operating officer of BIOCORP.
Easylog is a smart sensor that turns all classic pen injectors, disposable and reusable, into connected devices. Easylog captures key treatment information (dose, date, time) and transfers it to a monitoring mobile app. This solution goes hand in hand with a dedicated application that aims to facilitate the life of patients monitoring their treatment.
CE marking has been received for a version compatible with insulin delivery devices. Market launch is planned this through partnerships.
Founded in 2004 in Issoire (near Clermont-Ferrand), France, BIOCORP specializes in developing and manufacturing medical devices and drug delivery systems. With over twenty years of experience and more than 30 manufactured products, BIOCORP is a key player in the industry, providing drug delivery solutions that meet the evolving needs of patients. Today, BIOCORP continues to innovate in medical plastics, its core business, and to market traditional devices (alternative to aluminum capsules, syringe and vial administration systems) that have been an important source of recurring income. Its Internet-connected products include the DataPen, a reusable smart injection pen that automatically transmits data to a mobile app, helping patients to manage their treatment; and treatment management add-ons, which adapt to existing delivery devices and are compatible with most injection pens in the market. The company has a team of 48 employees.