Business Wire10.09.17
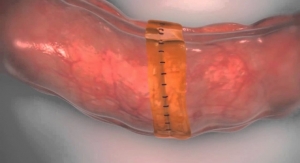
Amsel Medical Corporation has announced U.S. Food and Drug Administration 510(k) Pre-Marketing Notification clearance for its Low-Profile Amsel Occluder device.1 The Pre-Market Notification is the second 510(k) clearance2 received by the company for its family of Amsel Occluder devices. The Amsel Occluder device is intended for use in open general surgery procedures on tubular structures or vessels wherever a metal ligating clip is indicated and within the size range of 2 mm to 7 mm diameter.
Arnold Miller, M.D., founder and president of Amsel Medical Corporation commented, “We are thrilled about the clearance of the Pre-Market Notification 510(k) by FDA for the Low-Profile Amsel Occluder. The Low-Profile Amsel Occluder possesses the ability to transfix and ligate a selected vessel, advancing our technique beyond the current limitations of standard ligation clips. This simply applied device is the equivalent of a secure suture ligature, which will prevent loosening or slippage. We anticipate significant clinical interest for this technology in the markets where the Amsel Occluder is cleared for commercial distribution. This is another in a series of regulatory filings, while we continue the development of additional Amsel Occluder products.”
The Amsel Occluder device is designed to easily and securely close blood vessels (veins or arteries) as well as tubular structures during open surgical procedures. The Amsel Occluder is intended for use in open general surgery procedures on tubular structures or vessels wherever a metal ligating clip is indicated and within the size range of 2 mm to 7 mm diameter. The Amsel Occluder device employs a proprietary mechanical design that enables a vessel clamp to be introduced during open surgery. The Amsel Occluder device transfixes the target vessel with the clamp to provide closure.
Amsel Medical Corporation, a development stage medical device company founded in 2011, is developing the Amsel Occluder device, which is intended for simple and secure vessel or duct closure. The company has operations in Cambridge, Mass., and Tel Aviv, Israel. The Amsel Occluder Device addresses an estimated 31 million3,4 annual U.S. surgical procedures where vessel closure is required.
References
1. Patents pending.
2. On February 2, 2015, Amsel Medical Corporation announced its first Clearance of a 510(k) Pre-Marketing Notification by the US Food and Drug Administration for the Amsel Occluder Device.
3. CDC/NCHS National Hospital Discharge Survey, 2010.
4. Company estimates.
Arnold Miller, M.D., founder and president of Amsel Medical Corporation commented, “We are thrilled about the clearance of the Pre-Market Notification 510(k) by FDA for the Low-Profile Amsel Occluder. The Low-Profile Amsel Occluder possesses the ability to transfix and ligate a selected vessel, advancing our technique beyond the current limitations of standard ligation clips. This simply applied device is the equivalent of a secure suture ligature, which will prevent loosening or slippage. We anticipate significant clinical interest for this technology in the markets where the Amsel Occluder is cleared for commercial distribution. This is another in a series of regulatory filings, while we continue the development of additional Amsel Occluder products.”
The Amsel Occluder device is designed to easily and securely close blood vessels (veins or arteries) as well as tubular structures during open surgical procedures. The Amsel Occluder is intended for use in open general surgery procedures on tubular structures or vessels wherever a metal ligating clip is indicated and within the size range of 2 mm to 7 mm diameter. The Amsel Occluder device employs a proprietary mechanical design that enables a vessel clamp to be introduced during open surgery. The Amsel Occluder device transfixes the target vessel with the clamp to provide closure.
Amsel Medical Corporation, a development stage medical device company founded in 2011, is developing the Amsel Occluder device, which is intended for simple and secure vessel or duct closure. The company has operations in Cambridge, Mass., and Tel Aviv, Israel. The Amsel Occluder Device addresses an estimated 31 million3,4 annual U.S. surgical procedures where vessel closure is required.
References
1. Patents pending.
2. On February 2, 2015, Amsel Medical Corporation announced its first Clearance of a 510(k) Pre-Marketing Notification by the US Food and Drug Administration for the Amsel Occluder Device.
3. CDC/NCHS National Hospital Discharge Survey, 2010.
4. Company estimates.