PRWeb09.11.17
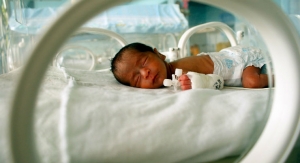
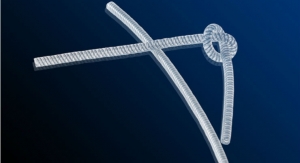
Using its innovative quick-disconnect technology, the SafeBreak Vascular helps to prevent IV failure and automatically alerts medical personnel when issues with the IV line occur.
Designed by Lineus Medical, SafeBreak Vascular is fitted with valves at either end of the device that stop the flow of fluid from the patient and the IV pump. When SafeBreak Vascular separates, the valves close and the pump signals that the IV line is in immediate need of attention.
Following its completion of the SafeBreak Vascular concept design phase, Lineus Medical recognized the need for a partner who could assist in executing the proper strategy, production planning, engineering, and manufacturing. For this, the company sought the expertise of BihlerMED in October of 2016, whose past experience in the medical device and contract manufacturing industries proved critical in the advancement of the SafeBreak Vascular breakaway technology.
Maxine Nordmeyer, CEO of Bihler of America, commented on the partnership, “BihlerMED’s mission is to join with leading edge companies to bring innovative products like SafeBreak Vascular to market that improve outcomes for doctors and patients alike. We view our relationship with Lineus Medical as an exciting opportunity to dramatically impact patient comfort and save lives.”
BihlerMED has considerable expertise in engineering, development, testing and manufacturing of medical devices. Its customer-specific, scalable automated production and assembly capabilities currently in development will help bring the SafeBreak Vascular to market, with product launch scheduled for Q1 2018.
Spencer Jones, founder and CEO of Lineus Medical, commented on the opportunity, “The worldwide market for peripheral IVs is over one billion units a year. We expect SafeBreak Vascular to grow very quickly, and we needed to find someone with whom we were confident could produce the parts to satisfy the demand. In BihlerMED, we believe we have that partner.”
Addressing Two Critical Issues
The development of the SafeBreak Vascular is aimed at improving a couple of growing problems—one medical, the other fiscal. Up to 90 percent of hospital patients receive intravenous (IV) medical treatment, and IVs prematurely fail on average 46 percent of the time. IV failure is painful for patients, time consuming for nurses and an expensive additional cost for healthcare systems. It is estimated that premature IV failure cost hospitals in the U.S. over $5 billion each year.
Reducing the Stress and Anxiety of IV Failure
BihlerMED has worked in close partnership with Lineus Medical to take the steps necessary to bring SafeBreak Vascular to market. It developed the strategic plan for the automated production, packaging, distribution and meeting the regulatory requirements of the product. BihlerMED’s on-site clean room research and development facility was invaluable in the design and engineering of the product’s packaging. BihlerMED has also designed the large-scale automated production for this product, which will be implemented to satisfy the demands of production and distribution.
Presently, Lineus Medical is completing the testing requirements required for FDA submission of SafeBreak Vascular, as BihlerMED is completing engineering and production design to bring the product effectively to market. Thanks to the partnership between BihlerMED and Lineus Medical, the considerable challenges associated with IV failure can be dramatically reduced, increasing patient comfort, decreasing the burden and stress on medical professionals, while saving hospitals countless dollars.
To learn more about BihlerMED’s services, including product support, design and engineering capabilities, visit BihlerMED at the 2017 MPO SUMMIT, October 18th and 19th in San Diego, Calif.
Designed by Lineus Medical, SafeBreak Vascular is fitted with valves at either end of the device that stop the flow of fluid from the patient and the IV pump. When SafeBreak Vascular separates, the valves close and the pump signals that the IV line is in immediate need of attention.
Following its completion of the SafeBreak Vascular concept design phase, Lineus Medical recognized the need for a partner who could assist in executing the proper strategy, production planning, engineering, and manufacturing. For this, the company sought the expertise of BihlerMED in October of 2016, whose past experience in the medical device and contract manufacturing industries proved critical in the advancement of the SafeBreak Vascular breakaway technology.
Maxine Nordmeyer, CEO of Bihler of America, commented on the partnership, “BihlerMED’s mission is to join with leading edge companies to bring innovative products like SafeBreak Vascular to market that improve outcomes for doctors and patients alike. We view our relationship with Lineus Medical as an exciting opportunity to dramatically impact patient comfort and save lives.”
BihlerMED has considerable expertise in engineering, development, testing and manufacturing of medical devices. Its customer-specific, scalable automated production and assembly capabilities currently in development will help bring the SafeBreak Vascular to market, with product launch scheduled for Q1 2018.
Spencer Jones, founder and CEO of Lineus Medical, commented on the opportunity, “The worldwide market for peripheral IVs is over one billion units a year. We expect SafeBreak Vascular to grow very quickly, and we needed to find someone with whom we were confident could produce the parts to satisfy the demand. In BihlerMED, we believe we have that partner.”
Addressing Two Critical Issues
The development of the SafeBreak Vascular is aimed at improving a couple of growing problems—one medical, the other fiscal. Up to 90 percent of hospital patients receive intravenous (IV) medical treatment, and IVs prematurely fail on average 46 percent of the time. IV failure is painful for patients, time consuming for nurses and an expensive additional cost for healthcare systems. It is estimated that premature IV failure cost hospitals in the U.S. over $5 billion each year.
Reducing the Stress and Anxiety of IV Failure
BihlerMED has worked in close partnership with Lineus Medical to take the steps necessary to bring SafeBreak Vascular to market. It developed the strategic plan for the automated production, packaging, distribution and meeting the regulatory requirements of the product. BihlerMED’s on-site clean room research and development facility was invaluable in the design and engineering of the product’s packaging. BihlerMED has also designed the large-scale automated production for this product, which will be implemented to satisfy the demands of production and distribution.
Presently, Lineus Medical is completing the testing requirements required for FDA submission of SafeBreak Vascular, as BihlerMED is completing engineering and production design to bring the product effectively to market. Thanks to the partnership between BihlerMED and Lineus Medical, the considerable challenges associated with IV failure can be dramatically reduced, increasing patient comfort, decreasing the burden and stress on medical professionals, while saving hospitals countless dollars.
To learn more about BihlerMED’s services, including product support, design and engineering capabilities, visit BihlerMED at the 2017 MPO SUMMIT, October 18th and 19th in San Diego, Calif.