Business Wire08.16.17
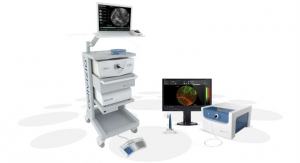
Mauna Kea Technologies, inventor of Cellvizio, the multidisciplinary confocal laser endomicroscopy (CLE) platform, has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its CelioFlex UHD Confocal Miniprobes for use with Cellvizio in robotic-assisted surgery procedures. The intended use of CelioFlex UHD Confocal Miniprobes is to provide visualization of body cavities, organs, and canals during endoscopic and laparoscopic surgical procedures, including robotic-assisted procedures. This is the 13th FDA clearance covering the Cellvizio system.
Robotic-assisted surgery allows doctors to perform many types of complex procedures with greater precision, flexibility and control than is possible with conventional techniques. There are more than 4,000 da Vinci robotic surgical systems installed worldwide, with approximately 750,000 robotic-assisted procedures performed on these systems in 2016.1 Robotic-assisted surgery has experienced significant growth in the last several years and is expected to continue growing rapidly as new robotic-assisted surgery systems enter the market.
Sacha Loiseau, Ph.D., founder and CEO of Mauna Kea Technologies, said, “This new FDA clearance is an important milestone for the company on its Cellvizio product roadmap and market expansion strategy. We see the ability to instantaneously visualize and characterize tissue at the cellular level as a natural extension to robotic-assisted procedures and enabling data-driven surgery, which will in-part rely on advanced imaging and machine learning algorithms to provide reliable, real-time tissue information to surgeons. ”
Mauna Kea Technologies is a global medical device company focused on eliminating uncertainties related to the diagnosis and treatment of cancer and other diseases thanks to real time in-vivo microscopic visualization. The company’s flagship product, Cellvizio, has received clearance to sell a wide range of applications in more than 40 countries, including the United States, Europe, Japan, China, Canada, Brazil and Mexico.
Reference
1. Intuitive Surgical Q2 2017 investor presentation
Robotic-assisted surgery allows doctors to perform many types of complex procedures with greater precision, flexibility and control than is possible with conventional techniques. There are more than 4,000 da Vinci robotic surgical systems installed worldwide, with approximately 750,000 robotic-assisted procedures performed on these systems in 2016.1 Robotic-assisted surgery has experienced significant growth in the last several years and is expected to continue growing rapidly as new robotic-assisted surgery systems enter the market.
Sacha Loiseau, Ph.D., founder and CEO of Mauna Kea Technologies, said, “This new FDA clearance is an important milestone for the company on its Cellvizio product roadmap and market expansion strategy. We see the ability to instantaneously visualize and characterize tissue at the cellular level as a natural extension to robotic-assisted procedures and enabling data-driven surgery, which will in-part rely on advanced imaging and machine learning algorithms to provide reliable, real-time tissue information to surgeons. ”
Mauna Kea Technologies is a global medical device company focused on eliminating uncertainties related to the diagnosis and treatment of cancer and other diseases thanks to real time in-vivo microscopic visualization. The company’s flagship product, Cellvizio, has received clearance to sell a wide range of applications in more than 40 countries, including the United States, Europe, Japan, China, Canada, Brazil and Mexico.
Reference
1. Intuitive Surgical Q2 2017 investor presentation