Business Wire07.19.17
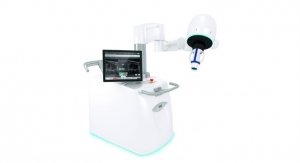
SCIEX Diagnostics, the in-vitro diagnostics division of SCIEX, a developer of mass spectrometry in the life sciences industry, said the AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system has been approved by the China Food and Drug Administration (CFDA) and meets the market access requirements for medical device products. With this accreditation, SCIEX aims to help propel China’s clinical laboratory programs to a new level by meeting the challenges of clinical diagnostic labs to deliver highly accurate and reliable test results.
The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system consists of a triple quadrupole mass spectrometer (TRIPLE QUADTM 4500MD), a high-performance liquid chromatography system (Jasper), and software including AnalystMD, CliquidMD and MultiQuantTMMD and it is intended to use with various reagents to analyze compounds in human specimens. The compounds include diagnostic indicators (endogenous substances: amino acids, vitamins, hormones) and therapeutic monitoring compounds (exogenous substances: therapeutic/toxic drugs).
The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system delivers excellent detection levels for the analytes commonly requested in clinical in-vitro diagnostic applications. Designed to maximize uptime in clinical laboratories and support continuous high-throughput operations, the AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system features the SCIEX Turbo V ion source and Curtain Gas interface for outstanding reliability, sensitivity and reproducibility in routine clinical testing and ensures day-to-day, month-to-month, or week-to-week measurement consistency. The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system provides scientists with the ability to perform multiple assays, and quantitate multiple compounds in a single run with a suitable reagent, resulting to fewer tests, significant time and cost savings in clinical laboratories.
“We are very pleased to share this news to with our customers and with the industry. We know that the routine diagnostic testing lab requires accurate trace level quantitation of multiple compounds in human specimens,” said Shao Hong, SCIEX China general manager. “The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system is a truly reliable platform, which can help meet clinical lab objectives, by saving time, cutting costs, and supporting physicians with greater confidence and reliable results. We believe that AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system will lead us to a new era in clinical in vitro diagnostic field.“
SCIEX helps scientists and laboratory analysts find answers to complex analytical challenges. The company's global leadership, service, and support in the capillary electrophoresis and liquid chromatography-mass spectrometry industry have made it a trusted partner to thousands of the scientists and lab analysts worldwide who are focused on basic research, drug discovery and development, food and environmental testing, forensics and clinical research. The firm's U.S. offices are located in Framingham, Mass.
The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system consists of a triple quadrupole mass spectrometer (TRIPLE QUADTM 4500MD), a high-performance liquid chromatography system (Jasper), and software including AnalystMD, CliquidMD and MultiQuantTMMD and it is intended to use with various reagents to analyze compounds in human specimens. The compounds include diagnostic indicators (endogenous substances: amino acids, vitamins, hormones) and therapeutic monitoring compounds (exogenous substances: therapeutic/toxic drugs).
The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system delivers excellent detection levels for the analytes commonly requested in clinical in-vitro diagnostic applications. Designed to maximize uptime in clinical laboratories and support continuous high-throughput operations, the AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system features the SCIEX Turbo V ion source and Curtain Gas interface for outstanding reliability, sensitivity and reproducibility in routine clinical testing and ensures day-to-day, month-to-month, or week-to-week measurement consistency. The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system provides scientists with the ability to perform multiple assays, and quantitate multiple compounds in a single run with a suitable reagent, resulting to fewer tests, significant time and cost savings in clinical laboratories.
“We are very pleased to share this news to with our customers and with the industry. We know that the routine diagnostic testing lab requires accurate trace level quantitation of multiple compounds in human specimens,” said Shao Hong, SCIEX China general manager. “The AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system is a truly reliable platform, which can help meet clinical lab objectives, by saving time, cutting costs, and supporting physicians with greater confidence and reliable results. We believe that AB SCIEX TRIPLE QUAD 4500MD LC-MS/MS system will lead us to a new era in clinical in vitro diagnostic field.“
SCIEX helps scientists and laboratory analysts find answers to complex analytical challenges. The company's global leadership, service, and support in the capillary electrophoresis and liquid chromatography-mass spectrometry industry have made it a trusted partner to thousands of the scientists and lab analysts worldwide who are focused on basic research, drug discovery and development, food and environmental testing, forensics and clinical research. The firm's U.S. offices are located in Framingham, Mass.