Business Wire06.30.17
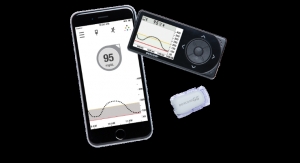
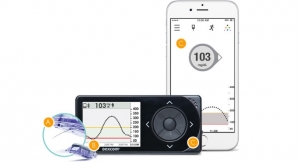
Tandem Diabetes Care Inc., manufacturer of the only touchscreen insulin pumps available in the United States, has released results from a feasibility study for its predictive low glucose suspend (PLGS) algorithm, designed to suspend insulin delivery when low blood sugar is predicted.
The overnight, hospital-based study recruited 10 subjects with type 1 diabetes ages 18 to 30 years old at the Barbara Davis Center for Diabetes and Stanford University. Participants started on the PLGS system in the evening, and low glucose was induced via increased basal insulin overnight. The system performed as expected during the study, successfully suspending insulin delivery when continuous glucose monitoring (CGM) values were predicted to be below 80 mg/dL in the next 30 minutes, and subsequently resuming insulin when CGM values began to rise. No hypoglycemic events were observed during the study, defined as reference glucose values <60 mg/dL, and peak CGM values two hours after suspension averaged 91 mg/dl, reflecting normal glycemic control without rebound hyperglycemia.
“The Tandem predictive low glucose suspend algorithm was extremely effective in anticipating low blood sugar and modulating insulin delivery accordingly,” said Gregory Forlenza, M.D., assistant professor of pediatrics at the Barbara Davis Center for Diabetes. “The touchscreen on the Tandem pump was simple to read and interpret, and this algorithm requires minimal interaction to operate. Based on this study, we are enthusiastic about the potential for this product, and we look forward to seeing data from the upcoming pivotal trial.”
“The results from this feasibility study are very encouraging. The IDE for our pivotal trial, which will use the predictive low glucose suspend algorithm on a t:slim X2 Pump with Dexcom G5 Mobile CGM integration, was approved by the FDA in May, and we look forward to starting enrollment soon,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “Our goal remains to submit our t:slim X2 Pump with predictive low glucose suspend to the FDA later this year, and we continue to plan for launch of this product in early 2018, subject to FDA approval.”
Tandem Diabetes Care Inc. is a medical device company with a user-centric and integrated approach to the design, development and commercialization of products for people with diabetes who use insulin. The company manufactures and sells the t:slim X2 Insulin Pump, the slimmest and smallest durable insulin pump currently on the market, the t:flex Insulin Pump, the first pump designed for people with greater insulin requirements, and the t:slim G4 Insulin Pump, the first continuous glucose monitoring-enabled pump with touchscreen simplicity. Tandem is based in San Diego, Calif.
The overnight, hospital-based study recruited 10 subjects with type 1 diabetes ages 18 to 30 years old at the Barbara Davis Center for Diabetes and Stanford University. Participants started on the PLGS system in the evening, and low glucose was induced via increased basal insulin overnight. The system performed as expected during the study, successfully suspending insulin delivery when continuous glucose monitoring (CGM) values were predicted to be below 80 mg/dL in the next 30 minutes, and subsequently resuming insulin when CGM values began to rise. No hypoglycemic events were observed during the study, defined as reference glucose values <60 mg/dL, and peak CGM values two hours after suspension averaged 91 mg/dl, reflecting normal glycemic control without rebound hyperglycemia.
“The Tandem predictive low glucose suspend algorithm was extremely effective in anticipating low blood sugar and modulating insulin delivery accordingly,” said Gregory Forlenza, M.D., assistant professor of pediatrics at the Barbara Davis Center for Diabetes. “The touchscreen on the Tandem pump was simple to read and interpret, and this algorithm requires minimal interaction to operate. Based on this study, we are enthusiastic about the potential for this product, and we look forward to seeing data from the upcoming pivotal trial.”
“The results from this feasibility study are very encouraging. The IDE for our pivotal trial, which will use the predictive low glucose suspend algorithm on a t:slim X2 Pump with Dexcom G5 Mobile CGM integration, was approved by the FDA in May, and we look forward to starting enrollment soon,” said Kim Blickenstaff, president and CEO of Tandem Diabetes Care. “Our goal remains to submit our t:slim X2 Pump with predictive low glucose suspend to the FDA later this year, and we continue to plan for launch of this product in early 2018, subject to FDA approval.”
Tandem Diabetes Care Inc. is a medical device company with a user-centric and integrated approach to the design, development and commercialization of products for people with diabetes who use insulin. The company manufactures and sells the t:slim X2 Insulin Pump, the slimmest and smallest durable insulin pump currently on the market, the t:flex Insulin Pump, the first pump designed for people with greater insulin requirements, and the t:slim G4 Insulin Pump, the first continuous glucose monitoring-enabled pump with touchscreen simplicity. Tandem is based in San Diego, Calif.