Tactile Systems Technology Inc. 06.01.17
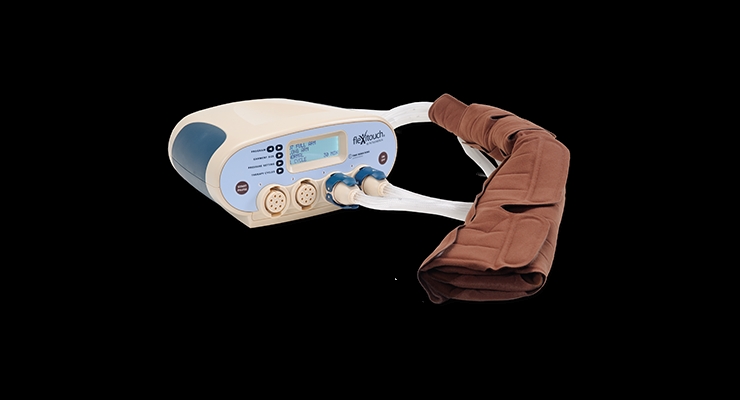
Tactile Systems Technology Inc., a developer and marketer of at-home therapy devices that treat lymphedema and chronic venous insufficiency, has launched its Flexitouch system throughout the United States to treat lymphedema of the head and neck, a common consequence of head and neck cancer and related treatment. The company claims Flexitouch is the first and only pneumatic compression therapy approved for this condition.
Following its U.S. Food and Drug Administration (FDA) clearance in September 2016, Tactile Medical completed a limited market release of the product, including more than 80 patients with lymphedema of the head and neck. Of the patients who tried the device, 88 percent saw a reduction in head and neck swelling after an initial 32-minute at-home treatment with the Flexitouch system, and 89 percent of patients reported improvement in other symptoms including increased ability to swallow, improved range of motion, a reduction in fibrotic (hardened) tissue and less discomfort.
“We are thrilled with these positive results demonstrating that the Flexitouch system can help head and neck lymphedema patients effectively reduce lymphedema swelling and improve their quality of life,” said Gerald R. Mattys, CEO of Tactile Medical. “The Flexitouch system has helped tens of thousands of patients achieve better health and manage lymphedema symptoms in their extremities. Now, for the first time, we can extend the benefits of Flexitouch to every area of the body.”
The Flexitouch system is the only pneumatic compression system clinically proven to stimulate the lymphatic system,1 delivering a non-invasive therapy that patients can administer themselves.
“We have been encouraged to hear that many patients are able to feel a substantial difference in their condition after just one treatment,” said Mattys. “With very few treatment options available for these patients, we are extremely gratified that we can now extend the benefits of our Flexitouch system to patients suffering from head and neck lymphedema and help them better manage their condition at home.”
Lymphedema is a chronic disease in which excess fluid accumulates in areas of the body when lymphatic vessels do not work efficiently or have been damaged. This chronic disease affects more than 5 million people in the United States. More than 75 percent of head and neck cancer patients experience lymphedema symptoms.2 While lymphedema is an incurable, chronic, and progressive condition, it can be effectively managed to relieve symptoms and lower the risk of skin damage and infection, reducing overall healthcare costs.
Tactile Medical develops and markets at-home therapy devices that treat lymphedema and chronic venous insufficiency. Its offering includes advanced, clinically proven pneumatic compression devices, as well as continuity of care services provided by a national network of product specialists and trainers, reimbursement experts, patient advocates, and clinical staff. This combination of products and services ensures that tens of thousands of patients annually receive the at-home treatment necessary to better manage their chronic conditions. The company is based in Minneapolis, Minn.
References
1. Adams KE, Rasmussen JC, Darne C, Tan I, Aldrich MB, Marshall MV, Fife CE, Maus EA, Smith LA, Guilloid R, Hoy S, Sevic-Muraca EM. Direct Evidence of Lymphatic Function Improvement After Advanced Pneumatic Compression Device Treatment of Lymphedema. Biomedical Optics Express. August 2010 (Vol 1/No 1).
2. Ridner, SH, Dietrich, MS, Niermann, K, Cmelak, A, Mannion, K, Murphy, B. A Prospective Study of the Lymphedema and Fibrosis Continuum in Patients with Head and Neck Cancer. Lymphatic Research and Biology. December 2016 (Vol 14/No 4).
Following its U.S. Food and Drug Administration (FDA) clearance in September 2016, Tactile Medical completed a limited market release of the product, including more than 80 patients with lymphedema of the head and neck. Of the patients who tried the device, 88 percent saw a reduction in head and neck swelling after an initial 32-minute at-home treatment with the Flexitouch system, and 89 percent of patients reported improvement in other symptoms including increased ability to swallow, improved range of motion, a reduction in fibrotic (hardened) tissue and less discomfort.
“We are thrilled with these positive results demonstrating that the Flexitouch system can help head and neck lymphedema patients effectively reduce lymphedema swelling and improve their quality of life,” said Gerald R. Mattys, CEO of Tactile Medical. “The Flexitouch system has helped tens of thousands of patients achieve better health and manage lymphedema symptoms in their extremities. Now, for the first time, we can extend the benefits of Flexitouch to every area of the body.”
The Flexitouch system is the only pneumatic compression system clinically proven to stimulate the lymphatic system,1 delivering a non-invasive therapy that patients can administer themselves.
“We have been encouraged to hear that many patients are able to feel a substantial difference in their condition after just one treatment,” said Mattys. “With very few treatment options available for these patients, we are extremely gratified that we can now extend the benefits of our Flexitouch system to patients suffering from head and neck lymphedema and help them better manage their condition at home.”
Lymphedema is a chronic disease in which excess fluid accumulates in areas of the body when lymphatic vessels do not work efficiently or have been damaged. This chronic disease affects more than 5 million people in the United States. More than 75 percent of head and neck cancer patients experience lymphedema symptoms.2 While lymphedema is an incurable, chronic, and progressive condition, it can be effectively managed to relieve symptoms and lower the risk of skin damage and infection, reducing overall healthcare costs.
Tactile Medical develops and markets at-home therapy devices that treat lymphedema and chronic venous insufficiency. Its offering includes advanced, clinically proven pneumatic compression devices, as well as continuity of care services provided by a national network of product specialists and trainers, reimbursement experts, patient advocates, and clinical staff. This combination of products and services ensures that tens of thousands of patients annually receive the at-home treatment necessary to better manage their chronic conditions. The company is based in Minneapolis, Minn.
References
1. Adams KE, Rasmussen JC, Darne C, Tan I, Aldrich MB, Marshall MV, Fife CE, Maus EA, Smith LA, Guilloid R, Hoy S, Sevic-Muraca EM. Direct Evidence of Lymphatic Function Improvement After Advanced Pneumatic Compression Device Treatment of Lymphedema. Biomedical Optics Express. August 2010 (Vol 1/No 1).
2. Ridner, SH, Dietrich, MS, Niermann, K, Cmelak, A, Mannion, K, Murphy, B. A Prospective Study of the Lymphedema and Fibrosis Continuum in Patients with Head and Neck Cancer. Lymphatic Research and Biology. December 2016 (Vol 14/No 4).