Business Wire05.04.17
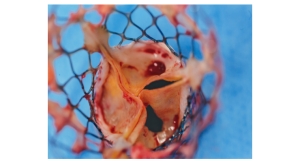
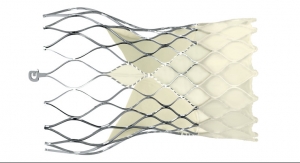
NaviGate Cardiac Structures Inc. (NCSI) has announced that its GATE catheter-guided tricuspid atrioventricular valved stent (AVS) was implanted through the jugular vein late last month in a patient suffering from severe tricuspid regurgitation stemming from two failed tricuspid annuloplasty rings that were unable to maintain the patient’s valve competence.
The patient, a 78-year-old man with a long history of cardiac conditions, has undergone four cardiac surgeries, including two coronary bypass surgeries and two failed valve repairs. The patient had become symptomatic of right heart failure and opted to make a compassionate plea to his physician for the valved stent under development. The U.S. Food and Drug Administration allowed the compassionate use. Dr. Jose Navia and Dr. Samir Kapadia proceeded with the implantation. After receiving the valve, the patient became stable. He has been closely watched by the cardiac team and was discharged home.
“We were able to perform this procedure in its entirety percutaneously through the jugular vein in a beating heart. In addition, the tricuspid valve was implanted within the failed annuloplasty ring, providing another alternative in treating tricuspid disease,” said Navia, the staff surgeon and researcher who sponsored the use at Cleveland Clinic. Navia, inventor of the technology, is a member of NaviGate’s scientific advisory board and a company shareholder.
“This was a patient who was very high-risk and would not have qualified for another conventional operation," explained Kapadia, director of the Catheterization Laboratory and member of the cardiac team at Cleveland Clinic. "Transcatheter valve implantation was performed in a hybrid room similar to other transcatheter valve replacements. The valve was implanted accurately and the patient has done well, and we will continue to monitor him throughout his recovery.” Kapadia is also a member of NaviGate’s scientific advisory board and holds equity in NCSI.
The GATE tricuspid AVS has been developed and manufactured by NCSI. The company also has developed the percutaneous NAVI valve for mitral valve replacement as well as tissue-preservation techniques that remove the toxic tissue fixatives thought to affect the longevity of tissue. This technology removes most of the liquid so that the device can be shipped in the “dry” form. NCSI licensed the seminal technology from Cleveland Clinic.
“Tricuspid valve regurgitation affects some 1 million patients in the U.S. and the treatment for heart failure related to the regurgitation can be difficult," said Dr. Lars Svensson, chair of the Miller Family Heart & Vascular Institute at Cleveland Clinic. "Jose Navia has had a long interest in the surgical treatment of the problem. Dr. Navia has now innovated an approach and for the first time has used a percutaneous approach through a neck vein to replace the valve. This is the type of pioneering that will make a difference in the treatment of this difficult condition.”
NCSI made modifications to the device that differentiates it from all others presently manufactured for atrioventricular valves. The design of this device in the form of a diffuser or truncated cone exhibits a low-height profile that can be more easily threaded through the vasculature to reach the atrioventricular valves, allowing it to reside without protrusion into either of the adjacent chambers (atrium or ventricle) for mitral or tricuspid valves, while simultaneously allowing the largest possible diameters to capture the enlarged annulus typical of the dysfunctional tricuspid valve resulting from Dilated Cardiomyopathy (DCM); thus, valves in sizes of up to 48 mm to 50 mm can be manufactured.
“We are so excited to see our device helping the patient so well,” said Dr. Tino Quijano, CEO of NCSI. “Although we tried the implantation in 3D reproductions of the patient’s right heart that included the non-circular annuloplasty ring and saw it work, when it is done in the patient it does bring to our heart a bit of anxiety, but we know that in the hands of the cardiac team, our device will correct the tricuspid incompetence and show safety and function. NCSI has obtained approvals in Chile and Poland to start implantations of the GATE AVS and patients are being screened. Its uniqueness has gained the attention of various cardiac centers in the Americas and Europe and as far as New Zealand, which are requesting it for so many patients for which now they hope to have a functioning alternative.”
The worldwide patient population for these two conditions in their severe stage is in the millions yearly, which explains the extensive effort being made by many large multinational medical companies as well as the plurality of start-up venture companies that have focused for the last decade on mitral replacement and repair and those that are now pursuing the repair and replacement of the “forgotten” tricuspid valve.
NaviGate Cardiac Structures Inc. is an early-stage company focused on developing transcatheter solutions for the less-invasive treatment of atrioventricular valve regurgitation. The NaviGate valved-base technology was licensed from Cleveland Clinic and further modified and developed by NCSI.
The patient, a 78-year-old man with a long history of cardiac conditions, has undergone four cardiac surgeries, including two coronary bypass surgeries and two failed valve repairs. The patient had become symptomatic of right heart failure and opted to make a compassionate plea to his physician for the valved stent under development. The U.S. Food and Drug Administration allowed the compassionate use. Dr. Jose Navia and Dr. Samir Kapadia proceeded with the implantation. After receiving the valve, the patient became stable. He has been closely watched by the cardiac team and was discharged home.
“We were able to perform this procedure in its entirety percutaneously through the jugular vein in a beating heart. In addition, the tricuspid valve was implanted within the failed annuloplasty ring, providing another alternative in treating tricuspid disease,” said Navia, the staff surgeon and researcher who sponsored the use at Cleveland Clinic. Navia, inventor of the technology, is a member of NaviGate’s scientific advisory board and a company shareholder.
“This was a patient who was very high-risk and would not have qualified for another conventional operation," explained Kapadia, director of the Catheterization Laboratory and member of the cardiac team at Cleveland Clinic. "Transcatheter valve implantation was performed in a hybrid room similar to other transcatheter valve replacements. The valve was implanted accurately and the patient has done well, and we will continue to monitor him throughout his recovery.” Kapadia is also a member of NaviGate’s scientific advisory board and holds equity in NCSI.
The GATE tricuspid AVS has been developed and manufactured by NCSI. The company also has developed the percutaneous NAVI valve for mitral valve replacement as well as tissue-preservation techniques that remove the toxic tissue fixatives thought to affect the longevity of tissue. This technology removes most of the liquid so that the device can be shipped in the “dry” form. NCSI licensed the seminal technology from Cleveland Clinic.
“Tricuspid valve regurgitation affects some 1 million patients in the U.S. and the treatment for heart failure related to the regurgitation can be difficult," said Dr. Lars Svensson, chair of the Miller Family Heart & Vascular Institute at Cleveland Clinic. "Jose Navia has had a long interest in the surgical treatment of the problem. Dr. Navia has now innovated an approach and for the first time has used a percutaneous approach through a neck vein to replace the valve. This is the type of pioneering that will make a difference in the treatment of this difficult condition.”
NCSI made modifications to the device that differentiates it from all others presently manufactured for atrioventricular valves. The design of this device in the form of a diffuser or truncated cone exhibits a low-height profile that can be more easily threaded through the vasculature to reach the atrioventricular valves, allowing it to reside without protrusion into either of the adjacent chambers (atrium or ventricle) for mitral or tricuspid valves, while simultaneously allowing the largest possible diameters to capture the enlarged annulus typical of the dysfunctional tricuspid valve resulting from Dilated Cardiomyopathy (DCM); thus, valves in sizes of up to 48 mm to 50 mm can be manufactured.
“We are so excited to see our device helping the patient so well,” said Dr. Tino Quijano, CEO of NCSI. “Although we tried the implantation in 3D reproductions of the patient’s right heart that included the non-circular annuloplasty ring and saw it work, when it is done in the patient it does bring to our heart a bit of anxiety, but we know that in the hands of the cardiac team, our device will correct the tricuspid incompetence and show safety and function. NCSI has obtained approvals in Chile and Poland to start implantations of the GATE AVS and patients are being screened. Its uniqueness has gained the attention of various cardiac centers in the Americas and Europe and as far as New Zealand, which are requesting it for so many patients for which now they hope to have a functioning alternative.”
The worldwide patient population for these two conditions in their severe stage is in the millions yearly, which explains the extensive effort being made by many large multinational medical companies as well as the plurality of start-up venture companies that have focused for the last decade on mitral replacement and repair and those that are now pursuing the repair and replacement of the “forgotten” tricuspid valve.
NaviGate Cardiac Structures Inc. is an early-stage company focused on developing transcatheter solutions for the less-invasive treatment of atrioventricular valve regurgitation. The NaviGate valved-base technology was licensed from Cleveland Clinic and further modified and developed by NCSI.