Biotronik SE & Co. KG02.22.17
Biotronik SE & Co. KG, a global developer of cardio- and endovascular medical technology, has developed an online tool that streamlines the workflow for physicians selecting the right MR-conditional heart monitor or pacing or ICD system for their patients.
The ProMRI Configurator can be found at www.promricheck.com and is an intuitive user-friendly tool that allows physicians to select the desired magnetic resonance imaging (MRI) conditions, subsequently generating a selection of all suitable magnetic resonance (MR)-conditional cardiac devices and leads approved and available in their country. In this way, physicians can ensure that patients receive the most appropriate MR-conditional cardiac system for access to MRI in case of a new implantation or a replacement.
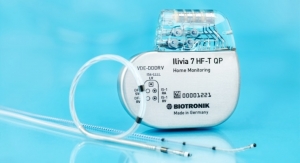
Biotronik manufactures a range of MR-conditional devices, along with a wide assortment of leads, offering a variety of options and combinations for different MR conditions. The ProMRI Configurator brings ease and convenience to the physician trying to find the most suitable set-up for the individual patients’ MRI needs.
The ProMRI Configurator marks the second offering of the ProMRI Check online platform. The first tool—the ProMRI SystemCheck—allows physicians and patients to retrospectively check whether an implanted cardiac system is MR-conditional. The ProMRI Configurator enhances the platform with its focus on the proactive selection of an MR-conditional system.
Last year, Biotronik won the Cardiostim Innovation Award in the category “Best Practice Improvement” for its MRI AutoDetect* feature in implantable cardioverter defibrillators, cardiac resynchronization devices and pacemakers. MRI AutoDetect recognizes an MRI environment and automatically switches to and from MRI mode without the cardiologist’s intervention. This not only eases the workflow for patients, physicians and radiologists, but also adds to patients’ safety.
“At Biotronik, we are invested in enabling as many patients as possible to have access to MRI scans,” saidd Manuel Ortega, senior vice president at Biotronik. “With the addition of the ProMRI Configurator to our portfolio, we are making further advances towards this goal by making it simpler for doctors to determine the best system for each patient.”
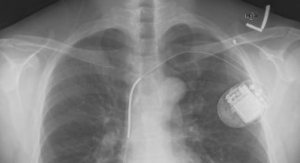
Biotronik ProMRI technology enables patients with a pacemaker, implantable defibrillator, cardiac monitor, or cardiac resynchronization therapy defibrillator (CRT-D) or pacemaker (CRT-P) to undergo an MRI scan. Devices with MRI AutoDetect have the additional capability of automatically recognizing an MR environment within a programmable time window, switching on the device’s MRI mode for only as long as is required to complete the scan, about 30 minutes. This ensures the patient receives the full benefit of their device for the maximum amount of time possible.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.
The ProMRI Configurator can be found at www.promricheck.com and is an intuitive user-friendly tool that allows physicians to select the desired magnetic resonance imaging (MRI) conditions, subsequently generating a selection of all suitable magnetic resonance (MR)-conditional cardiac devices and leads approved and available in their country. In this way, physicians can ensure that patients receive the most appropriate MR-conditional cardiac system for access to MRI in case of a new implantation or a replacement.
Biotronik manufactures a range of MR-conditional devices, along with a wide assortment of leads, offering a variety of options and combinations for different MR conditions. The ProMRI Configurator brings ease and convenience to the physician trying to find the most suitable set-up for the individual patients’ MRI needs.
The ProMRI Configurator marks the second offering of the ProMRI Check online platform. The first tool—the ProMRI SystemCheck—allows physicians and patients to retrospectively check whether an implanted cardiac system is MR-conditional. The ProMRI Configurator enhances the platform with its focus on the proactive selection of an MR-conditional system.
Last year, Biotronik won the Cardiostim Innovation Award in the category “Best Practice Improvement” for its MRI AutoDetect* feature in implantable cardioverter defibrillators, cardiac resynchronization devices and pacemakers. MRI AutoDetect recognizes an MRI environment and automatically switches to and from MRI mode without the cardiologist’s intervention. This not only eases the workflow for patients, physicians and radiologists, but also adds to patients’ safety.
“At Biotronik, we are invested in enabling as many patients as possible to have access to MRI scans,” saidd Manuel Ortega, senior vice president at Biotronik. “With the addition of the ProMRI Configurator to our portfolio, we are making further advances towards this goal by making it simpler for doctors to determine the best system for each patient.”
Biotronik ProMRI technology enables patients with a pacemaker, implantable defibrillator, cardiac monitor, or cardiac resynchronization therapy defibrillator (CRT-D) or pacemaker (CRT-P) to undergo an MRI scan. Devices with MRI AutoDetect have the additional capability of automatically recognizing an MR environment within a programmable time window, switching on the device’s MRI mode for only as long as is required to complete the scan, about 30 minutes. This ensures the patient receives the full benefit of their device for the maximum amount of time possible.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.