Business Wire02.21.17
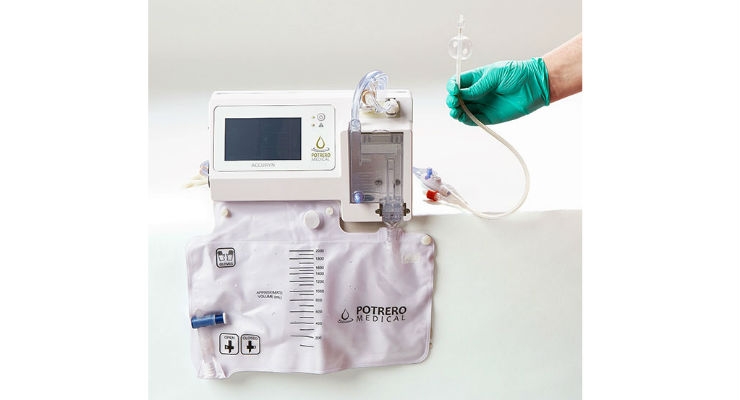
Medline Industries Inc., and Potrero Medical Inc., a privately held medical device company and the developer of the Accuryn critical care monitoring system, have announced a joint partnership to expand commercialization and continue clinical investigations to further support the benefits of the Accuryn critical care monitoring system. With this agreement, Medline has secured long-term exclusivity for marketing and distribution of Accuryn in North America. The ability to leverage Medline’s reach in the acute care hospital market ensures that as many critical care physicians and nurses as possible will have access to this system that empowers clinicians with more accurate and easily accessible data.
“We are pleased to partner with Medline to commercialize the world’s only technology capable of providing accurate, real-time data related to urine production and intra-abdominal pressure—the Accuryn Monitoring System,” said Dr. Daniel Burnett, CEO of Potrero Medical. “Medline’s reputation as a market leader combined with their customer base will make the Accuryn critical care monitoring system available to thousands of hospital systems and millions of patients in the United States.”
Accuryn is a critical care monitoring system that transforms the traditional urinary catheter into a next-generation diagnostic tool. Accuryn uses proprietary electromechanical designs and advanced software algorithms to deliver automated, real-time actionable data including urine output, core body temperature and intra-abdominal pressure (IAP). The IAP measurement is taken directly from the catheter tip with just a push of a button, helping to reduce setup time, potential errors, and infection risk. Additionally, innovative active drain line clearance can overcome urine drainage issues that may make urine output measurements unreliable.
“Medline is committed to being the leading supplier of clinically innovative solutions to healthcare providers. Our partnership with Potrero Medical on the Accuryn Monitoring System fully demonstrates this commitment and sets the pace for a new standard in critical care monitoring,” said David Greenberg, executive vice president of strategy and group president at Medline.
The Accuryn System is a U.S. Food and Drug Administration 510(k)-cleared medical device available for sale in the United States.
Medline is a global manufacturer and distributor serving the healthcare industry with medical supplies and clinical solutions. Headquartered in Northfield, Ill., the company offers more than 350,000 medical devices and support services through more than 1,200 direct sales representatives who are dedicated points of contact for customers across the continuum of care.
Potrero Medical, the latest spinout of medical device incubator Theranova LLC, is headquartered in San Francisco, Calif. and was founded with a mission to improve outcomes, reduce costs and expand access to healthcare.
“We are pleased to partner with Medline to commercialize the world’s only technology capable of providing accurate, real-time data related to urine production and intra-abdominal pressure—the Accuryn Monitoring System,” said Dr. Daniel Burnett, CEO of Potrero Medical. “Medline’s reputation as a market leader combined with their customer base will make the Accuryn critical care monitoring system available to thousands of hospital systems and millions of patients in the United States.”
Accuryn is a critical care monitoring system that transforms the traditional urinary catheter into a next-generation diagnostic tool. Accuryn uses proprietary electromechanical designs and advanced software algorithms to deliver automated, real-time actionable data including urine output, core body temperature and intra-abdominal pressure (IAP). The IAP measurement is taken directly from the catheter tip with just a push of a button, helping to reduce setup time, potential errors, and infection risk. Additionally, innovative active drain line clearance can overcome urine drainage issues that may make urine output measurements unreliable.
“Medline is committed to being the leading supplier of clinically innovative solutions to healthcare providers. Our partnership with Potrero Medical on the Accuryn Monitoring System fully demonstrates this commitment and sets the pace for a new standard in critical care monitoring,” said David Greenberg, executive vice president of strategy and group president at Medline.
The Accuryn System is a U.S. Food and Drug Administration 510(k)-cleared medical device available for sale in the United States.
Medline is a global manufacturer and distributor serving the healthcare industry with medical supplies and clinical solutions. Headquartered in Northfield, Ill., the company offers more than 350,000 medical devices and support services through more than 1,200 direct sales representatives who are dedicated points of contact for customers across the continuum of care.
Potrero Medical, the latest spinout of medical device incubator Theranova LLC, is headquartered in San Francisco, Calif. and was founded with a mission to improve outcomes, reduce costs and expand access to healthcare.