Owlstone Medical02.13.17
Owlstone Medical, a diagnostics company developing a breathalyzer for disease, has commenced a 1,400-patient clinical trial for the early detection of colorectal cancer using the company's FAIMS technology. Known as InTERCEPT, the trial aims to evaluate the accuracy of Owlstone Medical's quick, easy-to-use, non-invasive, high-compliance breath (and urine) test in the detection of colorectal cancer at an early stage, when patient survival rates are at their highest.
"A combination of low compliance and low sensitivity of current tests means too many patients are diagnosed when the cancer is at an advanced stage and survival chances are very poor. Two years ago my wife died of colorectal cancer as a direct result of late diagnosis," said Billy Boyle, co-founder and CEO at Owlstone Medical. "Early detection is our greatest opportunity for saving lives when chances of survival are higher than 90 percent. Through our InTERCEPT trial we hope to make this a reality for more patients."
Colorectal cancer is the second most common cancer killer worldwide, with 215,000 deaths every year at a cost of $99 billion. Current fecal screening tests are unpleasant and, as a result, have a low compliance rate. This, combined with relatively low test sensitivity particularly in early disease stages, means that too many patients are diagnosed with late stage disease, leading to poor patient outcomes.
"Early detection is crucial and any measures that increase the chances of cancer being picked up as quickly as possible are to be welcomed. Patients diagnosed with bowel cancer at the earliest stage have better than 90 percent chance of surviving for five years, whilst for those diagnosed at the latest stage this drops to just 6.6 percent," noted Dr. Fiona Reddington, head of Population, Prevention and Behavioural Research at CRUK. "However, there is considerable variation in participation levels in screening for bowel cancer cross the country, and the U.K. average is only 56 percent."
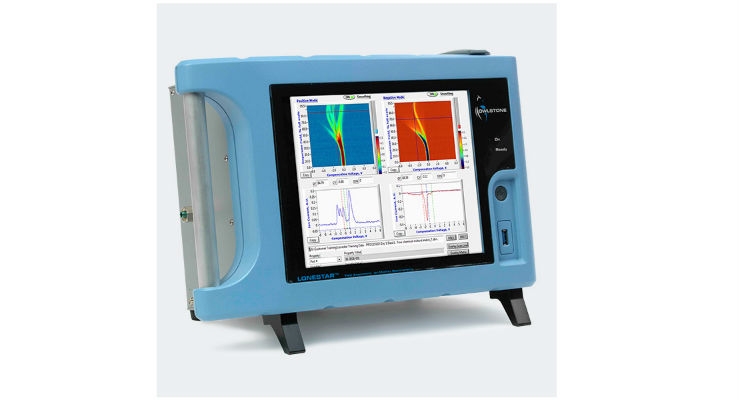
The InTERCEPT trial follows a successful pilot study using Owlstone Medical's microchip FAIMS (Field Asymmetric Ion Mobility Spectrometer) platform technology, which showed sensitivity of 88 percent in detecting Volatile Organic Compound (VOC) biomarkers for colorectal cancer. In addition, the pilot study showed sensitivity of 62 percent for detection of advanced adenomas, a pre-cancerous stage of colorectal cancer, representing a substantial increase in the rate of detection compared with the fecal occult blood tests based on detecting blood in feces that are used currently within the NHS bowel cancer screening program.
"My group has collaborated with Owlstone for several years in the discovery of biomarkers for a range of diseases including colorectal cancer, inflammatory bowel disease and tuberculosis. We are very impressed with the capability of Owlstone's new non-invasive technology and are excited to be working with them on this important trial.," said Professor Ramesh Arasaradnam, University of Warwick and consultant gastroenterologist at the University Hospitals Coventry Warwickshire NHS Trust.
The trial is being run in collaboration with the University of Warwick and the University Hospitals Coventry and Warwickshire NHS Trust with consultant gastroenterologist Arasaradnam as principal investigator for the study. The work has received widespread support from research organisations and charities including Bowel Cancer U.K., Cancer Research UK, the NCRI colorectal screening & prevention sub-group and the NIRI Clinical Research Network.
"The U.K. has had a bowel cancer screening program using stool testing for just over 10 years but it remains an unacceptable test to many people leading to low uptake," said Deborah Alsina MBE, chief executive of Bowel Cancer. "While pilot studies have shown this will improve with the introduction of the simpler fecal immunochemical test, an alternative to stool testing would be welcome both potentially as a population screening test and as a method primary care could use to triage people more effectively to further diagnostic testing such as a colonoscopy. Detecting bowel cancer early when treatment is more effective must remain our priority so we very much look forward to seeing the results of this innovative new approach to the earlier diagnosis of bowel cancer."
"There remains a clinical need for better diagnostic tests for colorectal cancer screening, prevention and early diagnosis, particularly modalities which may improve patient acceptability compared with stool testing. Owlstone Medical's clinical study is of high relevance to the NHS and its priorities. I look forward to supporting this important study," concluded Professor Mark Hull, a consultant gastroenterologist and chair of the NCRI colorectal screening & prevention sub-group.
Owlstone Medical's microchip FAIMS sensor is a platform technology that can be programmed in software to detect targeted biomarkers of disease in breath (and other biospecimens). FAIMS can be used in clinical diagnostics and precision medicine with application in cancer and a wide range of other medical conditions. Highly sensitive and selective, these tests allow for early diagnosis when treatments are more effective and where more lives can be saved.
Owlstone Medical is currently developing tests for lung and colorectal cancer, two of the most common cancer killers worldwide, and for asthma stratification by therapeutic response. The company also sells R&D tools and services to academic, clinical and pharma partners who want to develop breath based diagnostics for their own applications.
Founded in 2004 as a spin-out from the engineering department at the University of Cambridge University, Owlstone Inc is a profitable business selling its FAIMS technology to military and industrial customers globally. Owlstone Medical was spun out from Owlstone Inc. in 2016 to develop and commercialise FAIMS in diagnostic applications.
Owlstone Medical is headquartered in Cambridge, United Kingdom, with offices in London.
"A combination of low compliance and low sensitivity of current tests means too many patients are diagnosed when the cancer is at an advanced stage and survival chances are very poor. Two years ago my wife died of colorectal cancer as a direct result of late diagnosis," said Billy Boyle, co-founder and CEO at Owlstone Medical. "Early detection is our greatest opportunity for saving lives when chances of survival are higher than 90 percent. Through our InTERCEPT trial we hope to make this a reality for more patients."
Colorectal cancer is the second most common cancer killer worldwide, with 215,000 deaths every year at a cost of $99 billion. Current fecal screening tests are unpleasant and, as a result, have a low compliance rate. This, combined with relatively low test sensitivity particularly in early disease stages, means that too many patients are diagnosed with late stage disease, leading to poor patient outcomes.
"Early detection is crucial and any measures that increase the chances of cancer being picked up as quickly as possible are to be welcomed. Patients diagnosed with bowel cancer at the earliest stage have better than 90 percent chance of surviving for five years, whilst for those diagnosed at the latest stage this drops to just 6.6 percent," noted Dr. Fiona Reddington, head of Population, Prevention and Behavioural Research at CRUK. "However, there is considerable variation in participation levels in screening for bowel cancer cross the country, and the U.K. average is only 56 percent."
The InTERCEPT trial follows a successful pilot study using Owlstone Medical's microchip FAIMS (Field Asymmetric Ion Mobility Spectrometer) platform technology, which showed sensitivity of 88 percent in detecting Volatile Organic Compound (VOC) biomarkers for colorectal cancer. In addition, the pilot study showed sensitivity of 62 percent for detection of advanced adenomas, a pre-cancerous stage of colorectal cancer, representing a substantial increase in the rate of detection compared with the fecal occult blood tests based on detecting blood in feces that are used currently within the NHS bowel cancer screening program.
"My group has collaborated with Owlstone for several years in the discovery of biomarkers for a range of diseases including colorectal cancer, inflammatory bowel disease and tuberculosis. We are very impressed with the capability of Owlstone's new non-invasive technology and are excited to be working with them on this important trial.," said Professor Ramesh Arasaradnam, University of Warwick and consultant gastroenterologist at the University Hospitals Coventry Warwickshire NHS Trust.
The trial is being run in collaboration with the University of Warwick and the University Hospitals Coventry and Warwickshire NHS Trust with consultant gastroenterologist Arasaradnam as principal investigator for the study. The work has received widespread support from research organisations and charities including Bowel Cancer U.K., Cancer Research UK, the NCRI colorectal screening & prevention sub-group and the NIRI Clinical Research Network.
"The U.K. has had a bowel cancer screening program using stool testing for just over 10 years but it remains an unacceptable test to many people leading to low uptake," said Deborah Alsina MBE, chief executive of Bowel Cancer. "While pilot studies have shown this will improve with the introduction of the simpler fecal immunochemical test, an alternative to stool testing would be welcome both potentially as a population screening test and as a method primary care could use to triage people more effectively to further diagnostic testing such as a colonoscopy. Detecting bowel cancer early when treatment is more effective must remain our priority so we very much look forward to seeing the results of this innovative new approach to the earlier diagnosis of bowel cancer."
"There remains a clinical need for better diagnostic tests for colorectal cancer screening, prevention and early diagnosis, particularly modalities which may improve patient acceptability compared with stool testing. Owlstone Medical's clinical study is of high relevance to the NHS and its priorities. I look forward to supporting this important study," concluded Professor Mark Hull, a consultant gastroenterologist and chair of the NCRI colorectal screening & prevention sub-group.
Owlstone Medical's microchip FAIMS sensor is a platform technology that can be programmed in software to detect targeted biomarkers of disease in breath (and other biospecimens). FAIMS can be used in clinical diagnostics and precision medicine with application in cancer and a wide range of other medical conditions. Highly sensitive and selective, these tests allow for early diagnosis when treatments are more effective and where more lives can be saved.
Owlstone Medical is currently developing tests for lung and colorectal cancer, two of the most common cancer killers worldwide, and for asthma stratification by therapeutic response. The company also sells R&D tools and services to academic, clinical and pharma partners who want to develop breath based diagnostics for their own applications.
Founded in 2004 as a spin-out from the engineering department at the University of Cambridge University, Owlstone Inc is a profitable business selling its FAIMS technology to military and industrial customers globally. Owlstone Medical was spun out from Owlstone Inc. in 2016 to develop and commercialise FAIMS in diagnostic applications.
Owlstone Medical is headquartered in Cambridge, United Kingdom, with offices in London.