Fortimedix Surgical 10.07.16
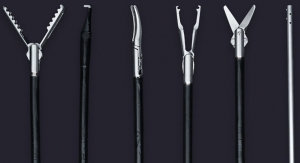
Fortimedix Surgical has received CE Mark approval for FMX314, the world’s first single-port surgery solution that is compatible with a standard 15 mm trocar for use in minimally invasive abdominal laparoscopic surgery. FMX314 addresses unmet needs in laparoscopic surgery by providing a platform solution that is small, simple and secure, according to the company.
“Our company is poised for significant growth, as we continue to achieve regulatory milestones to introduce innovative surgical instruments in key markets around the world,” said Marc van de Graaf, vice president of global business development of Fortimedix Surgical. “Following 510(k) clearance from the United States Food and Drug Administration last August, this CE Mark allows us to expand the availability of our innovative technology to patients in Europe, who will benefit from the unique advantages of FMX314.”
Because it is the first single-port platform that is compatible with a standard 15 mm laparoscopic trocar, FMX314 holds the promise of fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis compared with conventional multi-port laparoscopic surgery.
FMX314 emulates conventional, multi-port laparoscopy, making it easy to use and enabling surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
FMX314 will be launched in the United States during the American College of Surgeons Clinical Congress, taking place Oct. 16 – 20, 2016 in Washington, D.C. A European commercial launch is planned for 2017.
Fortimedix Surgical develops devices that capture the claimed benefits of single-port surgery. The company is headquartered in Nuth, The Netherlands, and has a U.S. subsidiary in San Diego, Calif.
“Our company is poised for significant growth, as we continue to achieve regulatory milestones to introduce innovative surgical instruments in key markets around the world,” said Marc van de Graaf, vice president of global business development of Fortimedix Surgical. “Following 510(k) clearance from the United States Food and Drug Administration last August, this CE Mark allows us to expand the availability of our innovative technology to patients in Europe, who will benefit from the unique advantages of FMX314.”
Because it is the first single-port platform that is compatible with a standard 15 mm laparoscopic trocar, FMX314 holds the promise of fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis compared with conventional multi-port laparoscopic surgery.
FMX314 emulates conventional, multi-port laparoscopy, making it easy to use and enabling surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
FMX314 will be launched in the United States during the American College of Surgeons Clinical Congress, taking place Oct. 16 – 20, 2016 in Washington, D.C. A European commercial launch is planned for 2017.
Fortimedix Surgical develops devices that capture the claimed benefits of single-port surgery. The company is headquartered in Nuth, The Netherlands, and has a U.S. subsidiary in San Diego, Calif.