PR Newswire08.30.16
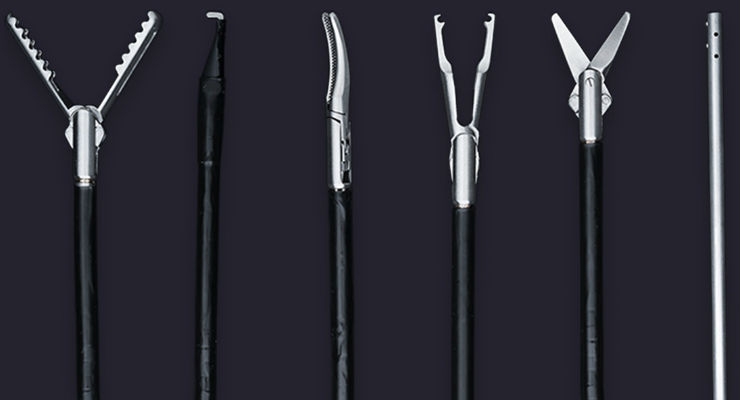
Fortimedix Surgical today (29 Aug) announced it has received 510(k) clearance from the US Food and Drug Administration (FDA) for FMX314, an innovative single-port platform for use in minimally invasive abdominal laparoscopic surgery.
"We are very pleased to announce we have secured 510(k) clearance for our platform, as it signifies an important milestone in our quest to deliver on the promise of single-port surgery," commented Mr. Wout Bijker, CEO of Fortimedix Surgical.
FMX314 is the world's first single-port surgery solution compatible with a standard 15mm laparoscopic trocar, promising fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis. Emulating conventional, multi-port laparoscopy makes FMX314 easy to use and enables surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
Market introduction in the United States is scheduled during the upcoming ACS Clinical Congress 2016 (October 16 - 20) in Washington, DC.
"We are very pleased to announce we have secured 510(k) clearance for our platform, as it signifies an important milestone in our quest to deliver on the promise of single-port surgery," commented Mr. Wout Bijker, CEO of Fortimedix Surgical.
FMX314 is the world's first single-port surgery solution compatible with a standard 15mm laparoscopic trocar, promising fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis. Emulating conventional, multi-port laparoscopy makes FMX314 easy to use and enables surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
Market introduction in the United States is scheduled during the upcoming ACS Clinical Congress 2016 (October 16 - 20) in Washington, DC.