PR Newswire07.20.16
Santen Pharmaceutical Co. Ltd. (Santen) is acquiring privately held InnFocus Inc., developer of the InnFocus MicroShunt glaucoma implant device.
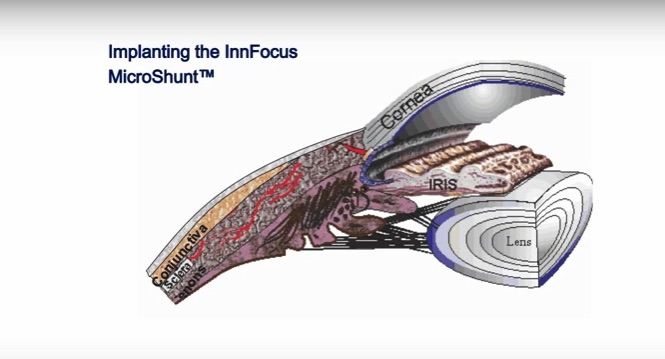
InnFocus is developing the MicroShunt implant to lower and sustain intraocular pressure (IOP) for the treatment of primary open-angle glaucoma (mild to severe stage disease). The MicroShunt has shown significant and sustainable lowering of IOP when used alone or in combination with cataract surgery in clinical trials outside of the United States. Late stage clinical studies are underway in the United States and Europe in advance of its pre-market approval application to the U.S. Food and Drug Administration (FDA). The MicroShunt has received CE Mark in Europe.
"With this acquisition, Santen will strengthen our glaucoma pipeline and stay at the forefront of innovation in ophthalmology. This agreement is in line with our long term vision to become a specialized pharmaceutical company with a global presence. I am truly excited about the MicroShunt as a new and effective treatment option that should significantly improve patient outcomes," said Akira Kurokawa, president and CEO of Santen.
Under the terms of the agreement, Santen will acquire InnFocus for $225 million, plus performance based consideration upon achievement of certain development, regulatory and commercial milestones. The acquisition is subject to certain conditions under the U.S. Hart-Scott-Rodino Antitrust Improvements Act.
"The InnFocus MicroShunt was developed to be the world's first minimally invasive stand-alone procedure for mild, moderate and severe stage primary open angle glaucoma. In addition to reducing IOP, the MicroShunt does not require simultaneous cataract removal, unlike many MIGS (micro-invasive glaucoma surgery) technologies. The InnFocus team is pleased to be joining Santen and its effort to address this growing and important segment of the ophthalmic market," said Randy Lindholm, executive board chairman of InnFocus.
The MicroShunt is a drainage implant device that helps effectively drain eye fluid and reduce intraocular pressure, which is the most effective means of preventing the progression of glaucoma and vision loss. The MicroShunt drainage device is made from SIBS; poly(styrene-block-isobutylene-block-styrene), a highly biocompatible biomaterial that has been implanted in the body for over 15 years. Compared to conventional glaucoma surgery, the MicroShunt is minimally invasive and anticipated to ease postoperative management.
Watch the video below to learn more about the MicroShunt:
InnFocus is an ophthalmology company founded in 2004 with extensive biomaterials experience that develops and provides next-generation products for glaucoma surgery. The MicroShunt is the first of many medical devices the company expects to incorporate biocompatible biomaterials in the eye. The MicroShunt was developed in collaboration with the University of Miami, Miller School of Medicine, Bascom Palmer Eye Institute.
Glaucoma is a disease in which the optic nerve (which transmits visual images to the brain) is damaged, thereby causing gradual vision loss. Elevated IOP is believed to be the main cause of optic nerve damage, and IOP lowering is considered to be the most effective treatment for preserving visual function.
As a specialty company dedicated to the ophthalmic field, Santen carries out research, development, sales, and marketing of pharmaceuticals. Santen is a provider in Japan for prescription ophthalmic pharmaceuticals and sells products in over 50 countries.
InnFocus is developing the MicroShunt implant to lower and sustain intraocular pressure (IOP) for the treatment of primary open-angle glaucoma (mild to severe stage disease). The MicroShunt has shown significant and sustainable lowering of IOP when used alone or in combination with cataract surgery in clinical trials outside of the United States. Late stage clinical studies are underway in the United States and Europe in advance of its pre-market approval application to the U.S. Food and Drug Administration (FDA). The MicroShunt has received CE Mark in Europe.
"With this acquisition, Santen will strengthen our glaucoma pipeline and stay at the forefront of innovation in ophthalmology. This agreement is in line with our long term vision to become a specialized pharmaceutical company with a global presence. I am truly excited about the MicroShunt as a new and effective treatment option that should significantly improve patient outcomes," said Akira Kurokawa, president and CEO of Santen.
Under the terms of the agreement, Santen will acquire InnFocus for $225 million, plus performance based consideration upon achievement of certain development, regulatory and commercial milestones. The acquisition is subject to certain conditions under the U.S. Hart-Scott-Rodino Antitrust Improvements Act.
"The InnFocus MicroShunt was developed to be the world's first minimally invasive stand-alone procedure for mild, moderate and severe stage primary open angle glaucoma. In addition to reducing IOP, the MicroShunt does not require simultaneous cataract removal, unlike many MIGS (micro-invasive glaucoma surgery) technologies. The InnFocus team is pleased to be joining Santen and its effort to address this growing and important segment of the ophthalmic market," said Randy Lindholm, executive board chairman of InnFocus.
The MicroShunt is a drainage implant device that helps effectively drain eye fluid and reduce intraocular pressure, which is the most effective means of preventing the progression of glaucoma and vision loss. The MicroShunt drainage device is made from SIBS; poly(styrene-block-isobutylene-block-styrene), a highly biocompatible biomaterial that has been implanted in the body for over 15 years. Compared to conventional glaucoma surgery, the MicroShunt is minimally invasive and anticipated to ease postoperative management.
Watch the video below to learn more about the MicroShunt:
InnFocus is an ophthalmology company founded in 2004 with extensive biomaterials experience that develops and provides next-generation products for glaucoma surgery. The MicroShunt is the first of many medical devices the company expects to incorporate biocompatible biomaterials in the eye. The MicroShunt was developed in collaboration with the University of Miami, Miller School of Medicine, Bascom Palmer Eye Institute.
Glaucoma is a disease in which the optic nerve (which transmits visual images to the brain) is damaged, thereby causing gradual vision loss. Elevated IOP is believed to be the main cause of optic nerve damage, and IOP lowering is considered to be the most effective treatment for preserving visual function.
As a specialty company dedicated to the ophthalmic field, Santen carries out research, development, sales, and marketing of pharmaceuticals. Santen is a provider in Japan for prescription ophthalmic pharmaceuticals and sells products in over 50 countries.