Viveve Medical Inc.07.08.16
Viveve Medical Inc., a medical technology company focused on women's health, has announced six new distribution partnerships that expand the commercial availability of the Viveve System to 14 additional countries in Latin America. The new relationships provide for distribution of the Viveve System in: Argentina, Peru, Paraguay (Sirex S.A.), Chile (Torregal), Columbia and Ecuador (Alphaeon Columbia S.A.S.), Mexico (Vitre-Tech), Panama, Costa Rica, Guatemala and the Dominican Republic (Coolmed, S.A.), Uruguay (Adenor, S.A.), and Venezuela and the Caribbean (MARC Group International, LLC).
"The Latin American region represents one of the world's largest markets for aesthetic medical procedures," said Jim Atkinson, president and chief business officer of Viveve. "Our collaboration with a distinguished group of distribution partners will help us to increase our presence in the region while providing outstanding service and customer support."
Viveve now has 23 exclusive partnerships covering distribution in 65 countries worldwide and has received regulatory clearance for the Viveve System in 25 of those countries thus far.
"We believe the combination of our internationally patented technology, the experience and professionalism of our partners, and the acceptance of innovative medical procedures in all of our target markets will advance Viveve's reputation as a provider of women's health products in this important region," said Patricia Scheller, CEO of Viveve. "Available safety and efficacy data continue to support the use of Viveve Treatment as a painless procedure to treat the common condition of vaginal laxity, which can profoundly impact a woman's sexual satisfaction and quality of life."
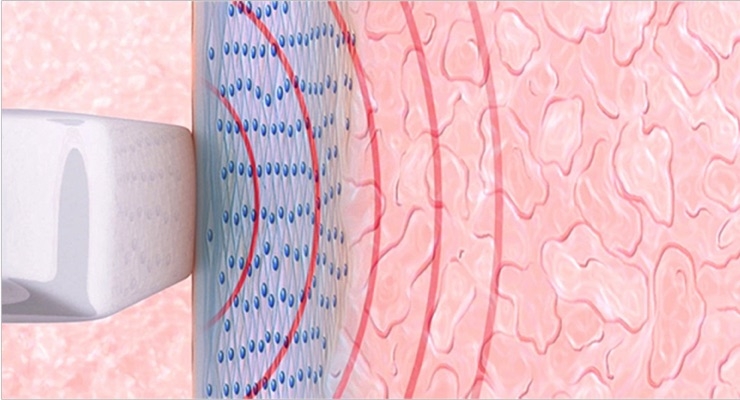
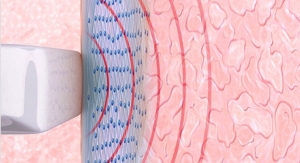
The company's lead product, the internationally patented Viveve System, is a non-surgical, non-ablative medical device that remodels collagen and restores tissue with only one treatment session. The Viveve System treats the condition of vaginal laxity that can result in decreased physical sensation and sexual satisfaction. Physician surveys indicate that vaginal laxity is the number one post-delivery physical change for women, being more prevalent than weight gain, urinary incontinence or stretch marks. The Viveve Treatment uses patented, reverse-thermal gradient radiofrequency technology to restore vaginal tissue in one 30-minute out-patient treatment in a physician's office. The Viveve System has received regulatory approval in many countries throughout the world and is available through physician import license in Japan. It is currently not available for sale in the United States.
Viveve Medical Inc., is a women's health company committed to advancing new solutions to improve women's overall well-being and quality of life. The firm is based in Sunnyvale, Calif.
"The Latin American region represents one of the world's largest markets for aesthetic medical procedures," said Jim Atkinson, president and chief business officer of Viveve. "Our collaboration with a distinguished group of distribution partners will help us to increase our presence in the region while providing outstanding service and customer support."
Viveve now has 23 exclusive partnerships covering distribution in 65 countries worldwide and has received regulatory clearance for the Viveve System in 25 of those countries thus far.
"We believe the combination of our internationally patented technology, the experience and professionalism of our partners, and the acceptance of innovative medical procedures in all of our target markets will advance Viveve's reputation as a provider of women's health products in this important region," said Patricia Scheller, CEO of Viveve. "Available safety and efficacy data continue to support the use of Viveve Treatment as a painless procedure to treat the common condition of vaginal laxity, which can profoundly impact a woman's sexual satisfaction and quality of life."
The company's lead product, the internationally patented Viveve System, is a non-surgical, non-ablative medical device that remodels collagen and restores tissue with only one treatment session. The Viveve System treats the condition of vaginal laxity that can result in decreased physical sensation and sexual satisfaction. Physician surveys indicate that vaginal laxity is the number one post-delivery physical change for women, being more prevalent than weight gain, urinary incontinence or stretch marks. The Viveve Treatment uses patented, reverse-thermal gradient radiofrequency technology to restore vaginal tissue in one 30-minute out-patient treatment in a physician's office. The Viveve System has received regulatory approval in many countries throughout the world and is available through physician import license in Japan. It is currently not available for sale in the United States.
Viveve Medical Inc., is a women's health company committed to advancing new solutions to improve women's overall well-being and quality of life. The firm is based in Sunnyvale, Calif.