Business Wire05.11.16
New clinical data from two randomized studies show that NeoTract Inc.'s UroLift System effectively treats patients with benign prostatic hyperplasia (BPH).
The conclusions were made based on four-year follow-up data from the pivotal, randomized L.I.F.T. IDE study, which evaluated the safety and effectiveness of the UroLift System in patients with symptomatic BPH, and two-year data from the BPH-6 study, which compared the UroLift System with the surgical standard, transurethral resection of the prostate (TURP).
“The clinical data show that treatment with the UroLift System offered men suffering from BPH a well-tolerated minimally invasive experience that provided uniquely rapid relief and a durable, sustained effect,” said Dave Amerson, president and CEO of NeoTract. “We are proud to offer this treatment option that can allow men to stop taking BPH medications and be treated with a simple procedure that does not carry the risk of sexual side effects common with other treatment options.”
Claus Roehrborn, M.D., professor and chair of the Department of Urology UT Southwestern Medical Center in Dallas, Texas, presented data from the L.I.F.T. IDE pivotal study at the American Urological Association's (AUA) 2016 Annual Meeting in San Diego, Calif. The data demonstrated durable improvement in symptoms, quality of life and urinary flow at four years.
“While prior data have shown that this treatment option offers rapid relief with low morbidity, the four-year L.I.F.T. study data demonstrate solid durability for a less invasive treatment option,” said Roerhborn, co-principal investigator for the L.I.F.T. clinical program. “After four years, over 86 percent of patients were free from seeking additional procedural treatment for BPH.”
The L.I.F.T. IDE Study enrolled 206 patients in a multi-center, randomized, blinded study to evaluate the safety and effectiveness of the UroLift System in patients with symptomatic BPH.
The four-year L.I.F.T. study analysis demonstrated that UroLift System treatment provided:
“While TURP is the most common treatment for symptoms caused by an enlarged prostate, the side effects can be significant and may cause men to postpone or refuse treatment altogether,” said Jens Sønksen, M.D., president of the Danish Association of Urology, who presented the data. “The results of this study provided an overall assessment of the ability of UroLift to improve symptoms without compromising important aspects of health, such as sexual function. We were also delighted to see that UroLift demonstrated superior quality of recovery. These characteristics are of great importance to patients when deciding how to address their symptoms.”
The BPH-6 study, a prospective, randomized study, compared the UroLift System treatment to TURP in 80 patients at 10 European centers, with a goal of evaluating the ability of the UroLift System to improve a patient's overall clinical improvement as compared to TURP.
Additional key data points from the study, at two years post-treatment:
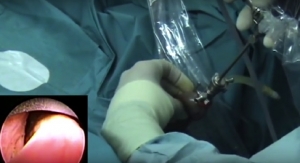
The U.S. Food and Drug Administration-cleared UroLift System is a minimally invasive technology for treating lower urinary tract symptoms due to benign prostatic hyperplasia (BPH). The UroLift permanent implants, delivered during a minimally invasive transurethral outpatient procedure, relieve prostate obstruction and open the urethra directly without cutting, heating, or removing prostate tissue. Clinical data from a pivotal 206-patient randomized controlled study showed that patients with enlarged prostate receiving UroLift implants reported rapid and durable symptomatic and urinary flow rate improvement without compromising sexual function. Patients also experienced a significant improvement in quality of life. Most common adverse events reported include hematuria, dysuria, micturition urgency, pelvic pain, and urge incontinence. Most symptoms were mild to moderate in severity and resolved within two to four weeks after the procedure. The UroLift System is available in the United States, Europe, Australia, Mexico, South Korea and Canada.
Watch the video below to learn more about the UroLift System for BPH:
NeoTract is dedicated to developing innovative, minimally invasive and clinically effective devices that address unmet needs in the field of urology. The Pleasanton, Calif.-based company’s initial focus is on improving the standard of care for patients with BPH using the UroLift System, a minimally invasive permanent implant system that treats symptoms while preserving normal sexual function.
The conclusions were made based on four-year follow-up data from the pivotal, randomized L.I.F.T. IDE study, which evaluated the safety and effectiveness of the UroLift System in patients with symptomatic BPH, and two-year data from the BPH-6 study, which compared the UroLift System with the surgical standard, transurethral resection of the prostate (TURP).
“The clinical data show that treatment with the UroLift System offered men suffering from BPH a well-tolerated minimally invasive experience that provided uniquely rapid relief and a durable, sustained effect,” said Dave Amerson, president and CEO of NeoTract. “We are proud to offer this treatment option that can allow men to stop taking BPH medications and be treated with a simple procedure that does not carry the risk of sexual side effects common with other treatment options.”
Claus Roehrborn, M.D., professor and chair of the Department of Urology UT Southwestern Medical Center in Dallas, Texas, presented data from the L.I.F.T. IDE pivotal study at the American Urological Association's (AUA) 2016 Annual Meeting in San Diego, Calif. The data demonstrated durable improvement in symptoms, quality of life and urinary flow at four years.
“While prior data have shown that this treatment option offers rapid relief with low morbidity, the four-year L.I.F.T. study data demonstrate solid durability for a less invasive treatment option,” said Roerhborn, co-principal investigator for the L.I.F.T. clinical program. “After four years, over 86 percent of patients were free from seeking additional procedural treatment for BPH.”
The L.I.F.T. IDE Study enrolled 206 patients in a multi-center, randomized, blinded study to evaluate the safety and effectiveness of the UroLift System in patients with symptomatic BPH.
The four-year L.I.F.T. study analysis demonstrated that UroLift System treatment provided:
- Rapid reduction of symptoms with low morbidity, while preserving sexual function;
- Sustained effect, with IPSS (International Prostate Symptom Score) and Qmax (peak urinary flow rate) remaining 41 percent and 62 percent improved from baseline, respectively;
- An improved quality of life (QoL), with the QoL score improved 52 percent over four years;
-
Freedom from retreatment for most patients, with 9 percent requiring alternative treatment and 4 percent requiring additional UroLift implants over the course of four years.
“While TURP is the most common treatment for symptoms caused by an enlarged prostate, the side effects can be significant and may cause men to postpone or refuse treatment altogether,” said Jens Sønksen, M.D., president of the Danish Association of Urology, who presented the data. “The results of this study provided an overall assessment of the ability of UroLift to improve symptoms without compromising important aspects of health, such as sexual function. We were also delighted to see that UroLift demonstrated superior quality of recovery. These characteristics are of great importance to patients when deciding how to address their symptoms.”
The BPH-6 study, a prospective, randomized study, compared the UroLift System treatment to TURP in 80 patients at 10 European centers, with a goal of evaluating the ability of the UroLift System to improve a patient's overall clinical improvement as compared to TURP.
Additional key data points from the study, at two years post-treatment:
- UroLift provided superior quality of recovery and preservation of sexual function as compared to TURP, p<0.01.
- Both UroLift and TURP provided significant symptom relief, quality of life and flow improvement through two years.
- While improvement in International IPSS and Qmax were statistically superior for TURP, this did not result in a difference in quality of life improvement.
-
The need for additional treatment, whether for LUTS or complications, was the same between groups, 14 percent over two years.
The U.S. Food and Drug Administration-cleared UroLift System is a minimally invasive technology for treating lower urinary tract symptoms due to benign prostatic hyperplasia (BPH). The UroLift permanent implants, delivered during a minimally invasive transurethral outpatient procedure, relieve prostate obstruction and open the urethra directly without cutting, heating, or removing prostate tissue. Clinical data from a pivotal 206-patient randomized controlled study showed that patients with enlarged prostate receiving UroLift implants reported rapid and durable symptomatic and urinary flow rate improvement without compromising sexual function. Patients also experienced a significant improvement in quality of life. Most common adverse events reported include hematuria, dysuria, micturition urgency, pelvic pain, and urge incontinence. Most symptoms were mild to moderate in severity and resolved within two to four weeks after the procedure. The UroLift System is available in the United States, Europe, Australia, Mexico, South Korea and Canada.
Watch the video below to learn more about the UroLift System for BPH:
NeoTract is dedicated to developing innovative, minimally invasive and clinically effective devices that address unmet needs in the field of urology. The Pleasanton, Calif.-based company’s initial focus is on improving the standard of care for patients with BPH using the UroLift System, a minimally invasive permanent implant system that treats symptoms while preserving normal sexual function.