Business Wire04.14.16
Acelity, a global advanced wound care and regenerative medicine company, announced that its Tielle foam dressing Family is now available in the United States. The TielleI dressing family expands the advanced wound therapeutics portfolio from Acelity.
This expansion complements the company's existing solutions for chronic and acute wounds and provides clinicians with efficient and cost-effective options for treating patients.
“Our goal was to reinvigorate the range of Tielle dressings and introduce it to clinicians here in the U.S. who are seeking effective solutions for all patients, including the most compromised,” said Joe Woody, president and CEO of Acelity. “We aim to strengthen our partnership with nurses and physicians by broadening our platform to include an extensive range of dressings, advancements in negative pressure technology and new innovations designed to help speed healing for patients and reduce the overall cost of care.”
The Tielle foam dressing family was designed to meet the needs of healthcare professionals, including exudate management and reduced pain for the patient at dressing removal.
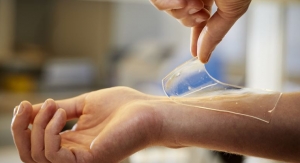
The Tielle Hydropolymer dressings – Tielle Silicone border and Tielle Non-Adhesive – include the Liqualock Advanced Absorption technology, which locks fluid away to help prevent leakage and maceration, according to Acelity. Both dressings are intended to manage a range of exudate levels in partial or full thickness wounds. They are designed to help maintain a moist wound healing environment through the combination of fluid absorption and moisture vapor transfer rate (MVTR), and to be worn for up to seven days depending on the wound condition and exudate levels, which may help reduce the number of dressing changes and associated cost, a benefit for patients, payors and providers.
Systagenix, an Acelity company, introduced the Tielle Hydropolymer dressing range in Europe in 1992. In 2013, KCI commercialized negative pressure wound therapy with the introduction of the V.A.C. therapy system in 1995 and then strengthened its position in the global wound therapeutics industry by acquiring Systagenix in 2012, increasing the company’s ability to offer best-in-class products across the wound care continuum. In 2014, KCI, Systagenix, and LifeCell, a developer of tissue repair products, combined to form Acelity, a global corporation devoted to the art and science of healing. Headquartered in San Antonio, Texas, Acelity employs more than 5,800 people around the world.
This expansion complements the company's existing solutions for chronic and acute wounds and provides clinicians with efficient and cost-effective options for treating patients.
“Our goal was to reinvigorate the range of Tielle dressings and introduce it to clinicians here in the U.S. who are seeking effective solutions for all patients, including the most compromised,” said Joe Woody, president and CEO of Acelity. “We aim to strengthen our partnership with nurses and physicians by broadening our platform to include an extensive range of dressings, advancements in negative pressure technology and new innovations designed to help speed healing for patients and reduce the overall cost of care.”
The Tielle foam dressing family was designed to meet the needs of healthcare professionals, including exudate management and reduced pain for the patient at dressing removal.
The Tielle Hydropolymer dressings – Tielle Silicone border and Tielle Non-Adhesive – include the Liqualock Advanced Absorption technology, which locks fluid away to help prevent leakage and maceration, according to Acelity. Both dressings are intended to manage a range of exudate levels in partial or full thickness wounds. They are designed to help maintain a moist wound healing environment through the combination of fluid absorption and moisture vapor transfer rate (MVTR), and to be worn for up to seven days depending on the wound condition and exudate levels, which may help reduce the number of dressing changes and associated cost, a benefit for patients, payors and providers.
- Tielle Silicone Border is intended to manage a range of exudate levels in partial or full thickness wounds. The soft silicone adhesive spans the full extent of the dressing, allowing for gentle removal, helping to minimize pain and trauma for the patient.
-
Tielle Non-Adhesive is a low profile dressing, which supports patient comfort and may be cut to size.
- Tielle Essential Non-Adhesive and Heel
- Tielle Essential Border
- Tielle Essential Silicone Border
- Tielle Essential Silicone Border Sacrum
-
Tielle Essential Silicone
Systagenix, an Acelity company, introduced the Tielle Hydropolymer dressing range in Europe in 1992. In 2013, KCI commercialized negative pressure wound therapy with the introduction of the V.A.C. therapy system in 1995 and then strengthened its position in the global wound therapeutics industry by acquiring Systagenix in 2012, increasing the company’s ability to offer best-in-class products across the wound care continuum. In 2014, KCI, Systagenix, and LifeCell, a developer of tissue repair products, combined to form Acelity, a global corporation devoted to the art and science of healing. Headquartered in San Antonio, Texas, Acelity employs more than 5,800 people around the world.