Aytu Bioscience Inc. 04.04.16
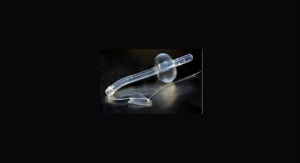
Aytu BioScience Inc. has obtained Health Canada Class II Medical Device approval for its MiOXSYS System. MiOXSYS is the company’s in-vitro diagnostic platform for assessing the level of oxidative stress in semen as an aid in the diagnosis of infertility in males.
The MiOXSYS System received CE Marking in the European Union in January and has posted its first commercial sales in Europe and the Middle East. With Health Canada approval now in place, the company will begin initial marketing of the product, while also looking for strategic opportunities to build partnerships with prominent hospitals, academic centers, and early MiOXSYS users to develop the market in Canada.
“Achievement of this milestone reflects our focus on commercializing best-in-class urology products and marketing those products strategically," said Josh Disbrow, CEO. "As we pursue FDA [U.S. Food and Drug Administration] clearance of MiOXSYS in the United States, we expect to benefit greatly from these early commercialization efforts in key markets. In Europe and the Middle East, our urology customers are already beginning to see the value of MiOXSYS, and this interest is being driven by the widespread need for improved diagnosis in the field of male infertility.”
Aytu BioScience is a commercial-stage specialty healthcare company that develops techologies for treatment of urological conditions. Aytu’s current portfolio of commercial and late-stage urology products addresses prostate cancer, urinary tract infections, male infertility and male sexual dysfunction. The Englewood, Colo.-based company currently markets ProstaScint (capromab pendetide), the only radio-labeled monoclonal antibody that targets prostate specific membrane antigen, a protein highly expressed by prostate cancer cells. ProstaScint is U.S. Food and Drug Administration (FDA)-approved as an imaging agent for use in both newly diagnosed, high-risk prostate cancer patients and patients with recurrent prostate cancer. Aytu also markets Primsol (trimethoprim hydrochloride) – the only FDA-approved trimethoprim-only oral solution for urinary tract infections. Additionally, Aytu markets the CE Marked MiOXSYS System outside the United States and is conducting U.S.-based clinical trials in the hopes of receiving 510(k) de novo medical device clearance. The MiOXSYS System is a rapid semen analysis system for assessing oxidative stress in semen and seminal plasma, a leading contributor of idiopathic male infertility.
The MiOXSYS System received CE Marking in the European Union in January and has posted its first commercial sales in Europe and the Middle East. With Health Canada approval now in place, the company will begin initial marketing of the product, while also looking for strategic opportunities to build partnerships with prominent hospitals, academic centers, and early MiOXSYS users to develop the market in Canada.
“Achievement of this milestone reflects our focus on commercializing best-in-class urology products and marketing those products strategically," said Josh Disbrow, CEO. "As we pursue FDA [U.S. Food and Drug Administration] clearance of MiOXSYS in the United States, we expect to benefit greatly from these early commercialization efforts in key markets. In Europe and the Middle East, our urology customers are already beginning to see the value of MiOXSYS, and this interest is being driven by the widespread need for improved diagnosis in the field of male infertility.”
Aytu BioScience is a commercial-stage specialty healthcare company that develops techologies for treatment of urological conditions. Aytu’s current portfolio of commercial and late-stage urology products addresses prostate cancer, urinary tract infections, male infertility and male sexual dysfunction. The Englewood, Colo.-based company currently markets ProstaScint (capromab pendetide), the only radio-labeled monoclonal antibody that targets prostate specific membrane antigen, a protein highly expressed by prostate cancer cells. ProstaScint is U.S. Food and Drug Administration (FDA)-approved as an imaging agent for use in both newly diagnosed, high-risk prostate cancer patients and patients with recurrent prostate cancer. Aytu also markets Primsol (trimethoprim hydrochloride) – the only FDA-approved trimethoprim-only oral solution for urinary tract infections. Additionally, Aytu markets the CE Marked MiOXSYS System outside the United States and is conducting U.S.-based clinical trials in the hopes of receiving 510(k) de novo medical device clearance. The MiOXSYS System is a rapid semen analysis system for assessing oxidative stress in semen and seminal plasma, a leading contributor of idiopathic male infertility.