03.29.16
Fortimedix Surgical said it has submitted a 510(k) approval application for its FMX314 single-port surgery solution with the U.S. Food and Drug Administration (FDA).
“We are very pleased to announce our submission with the FDA, as it represents an important milestone towards market introduction of FMX314 in the United States, which is expected in the fourth quarter of this year,” said Marc van de Graaf, vice president of global business development for Fortimedix Surgical.
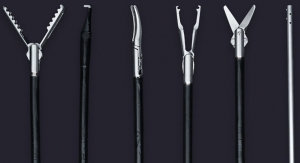
FMX314 is the world’s first single-port surgery solution that is compatible with a standard 15mm laparoscopic trocar, promising fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis, according to the company. Closely mimicking conventional, multi-port laparoscopy makes FMX314 easy to use and enables surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
“With its unique sizing and ease-of-use, we believe FMX314 will deliver on the promise of single-port surgery, elevating the standard of care in laparoscopy,” said Van de Graaf.
The FMX314 Surgical Platform is an investigational device only and not available for sale.
Fortimedix Surgical is a developer of devices that capture the claimed benefits of single-port surgery. The company is headquartered in Nuth, The Netherlands, and has a U.S. subsidiary in San Diego, Calif.
“We are very pleased to announce our submission with the FDA, as it represents an important milestone towards market introduction of FMX314 in the United States, which is expected in the fourth quarter of this year,” said Marc van de Graaf, vice president of global business development for Fortimedix Surgical.
FMX314 is the world’s first single-port surgery solution that is compatible with a standard 15mm laparoscopic trocar, promising fewer port-site complications, less post-operative pain, faster recovery and exceptional cosmesis, according to the company. Closely mimicking conventional, multi-port laparoscopy makes FMX314 easy to use and enables surgeons to perform procedural steps ergonomically, allowing for a comfortable and secure single-port approach.
“With its unique sizing and ease-of-use, we believe FMX314 will deliver on the promise of single-port surgery, elevating the standard of care in laparoscopy,” said Van de Graaf.
The FMX314 Surgical Platform is an investigational device only and not available for sale.
Fortimedix Surgical is a developer of devices that capture the claimed benefits of single-port surgery. The company is headquartered in Nuth, The Netherlands, and has a U.S. subsidiary in San Diego, Calif.