Cardica Inc. 01.08.16
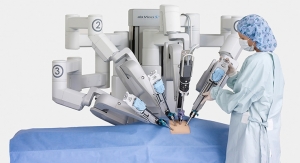
Cardica Inc. has amended its license agreement, initially signed in August 2010, with Intuitive Surgical Inc. to include an agreement providing for the evaluation and potential development of a surgical stapling cartridge for use with Intuitive Surgical’s da Vinci Surgical Systems.
Under the terms of the amendment, Intuitive Surgical has paid Cardica $2 million to extend its rights to improvements in Cardica’s stapling technology and certain patents until August 2018 and to provide for a feasibility evaluation period of up to six months.
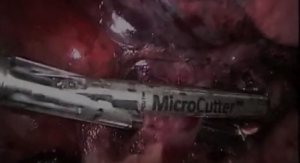
The feasibility evaluation allows Intuitive Surgical to test and evaluate Cardica’s MicroCutter XCHANGE technology. Upon completion of the evaluation, Intuitive Surgical will have the option to initiate a joint development program, for an 8 mm in diameter surgical stapling cartridge for use with the da Vinci Surgical System. Cardica may receive further funding for development of the cartridge and tooling as well as a unit-based royalty on commercial sales.
“We believe the relationship with Intuitive Surgical highlights two innovative technologies coming together to facilitate less invasive surgical procedures, for the ultimate benefit of surgeons and their patients,” said Julian Nikolchev, president/CEO of Cardica. “Importantly, with our new management team in place, this amendment to our agreement with Intuitive Surgical aligns with our plans to extend the MicroCutter product line while maintaining internal focus on completing the development of the MicroCutter XCHANGE 30.”
Based in Redwood City, Calif., Cardica designs and manufactures proprietary stapling and anastomotic devices for cardiac and laparoscopic surgical procedures. Cardica's technology portfolio is intended to reduce operating time and facilitate minimally-invasive and robot-assisted surgeries. Cardica’s MicroCutter XCHANGE 30, includes a cartridge-based articulating surgical stapling device with a 5-mm shaft diameter and staples, is manufactured and cleared for use in the United States for multiple open or minimally invasive surgical procedures for the transection, resection, and/or creation of anastomoses in small and large intestine as well as the transection of the appendix. In Europe the device has applications in multiple general, gynecologic, urologic, thoracic and pediatric surgical procedures. In addition, Cardica manufactures and markets its automated anastomosis systems, the C-Port Distal Anastomosis Systems and PAS-Port Proximal Anastomosis System for coronary artery bypass graft (CABG) surgery, and has shipped over 56,500 units throughout the world.
Under the terms of the amendment, Intuitive Surgical has paid Cardica $2 million to extend its rights to improvements in Cardica’s stapling technology and certain patents until August 2018 and to provide for a feasibility evaluation period of up to six months.
The feasibility evaluation allows Intuitive Surgical to test and evaluate Cardica’s MicroCutter XCHANGE technology. Upon completion of the evaluation, Intuitive Surgical will have the option to initiate a joint development program, for an 8 mm in diameter surgical stapling cartridge for use with the da Vinci Surgical System. Cardica may receive further funding for development of the cartridge and tooling as well as a unit-based royalty on commercial sales.
“We believe the relationship with Intuitive Surgical highlights two innovative technologies coming together to facilitate less invasive surgical procedures, for the ultimate benefit of surgeons and their patients,” said Julian Nikolchev, president/CEO of Cardica. “Importantly, with our new management team in place, this amendment to our agreement with Intuitive Surgical aligns with our plans to extend the MicroCutter product line while maintaining internal focus on completing the development of the MicroCutter XCHANGE 30.”
Based in Redwood City, Calif., Cardica designs and manufactures proprietary stapling and anastomotic devices for cardiac and laparoscopic surgical procedures. Cardica's technology portfolio is intended to reduce operating time and facilitate minimally-invasive and robot-assisted surgeries. Cardica’s MicroCutter XCHANGE 30, includes a cartridge-based articulating surgical stapling device with a 5-mm shaft diameter and staples, is manufactured and cleared for use in the United States for multiple open or minimally invasive surgical procedures for the transection, resection, and/or creation of anastomoses in small and large intestine as well as the transection of the appendix. In Europe the device has applications in multiple general, gynecologic, urologic, thoracic and pediatric surgical procedures. In addition, Cardica manufactures and markets its automated anastomosis systems, the C-Port Distal Anastomosis Systems and PAS-Port Proximal Anastomosis System for coronary artery bypass graft (CABG) surgery, and has shipped over 56,500 units throughout the world.