Kim Rahilly11.13.15
Proof-of-concept of the Wearable Artificial Kidney (WAK), the externally worn continuous-dialysis device on a belt, was confirmed with the third successful human clinical trial, this one in the United States, following those conducted in Italy and the U.K.
Nephrologist and inventor of the WAK Victor Gura M.D., who is affiliated with both UCLA and Cedars Sinai Medical Center, presented the results Saturday at the American Society of Nephrology’s annual ASN Kidney Week conference in San Diego, California. The WAK represents the first material advancement in dialysis technology in decades offering new hope for the millions of hemodialysis patients around the globe.
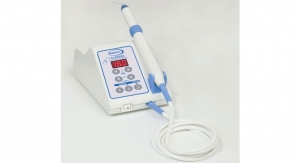
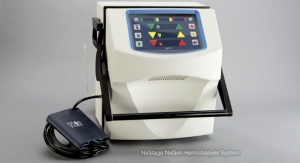
The WAK, which stands for Wearable Artificial Kidney, is a belt-like device that miniaturizes the functions of a 300-pound dialysis machine down to 10 pounds, and the WAK research team believes they can reduce its weight to half that.
It uses just a half liter of water as opposed to 40 gallons per session, and is battery-operated, user-friendly and lightweight allowing for freedom of movement for patients. Much like a normal kidney, the WAK removes excess fluids and toxins from the blood continuously at a natural rate, allowing them to achieve a quality of life closer to that of a healthy person.
Traditional dialysis makes a normal life impossible for patients not only because they need to be strapped to a kidney dialysis machine for 9-12 hours per week but because it severely restricts their diets. With the WAK, patients can enjoy most of the foods forbidden to them for years; improve their quality of life; reduce hospitalizations and increase longevity. Currently, the mortality rate for ESRD patients in the U.S. is at an unacceptable 20%.
Seven patients in Seattle, WA who have end-stage kidney disease (ESRD), took part in the study and were fitted with the device. The patients were encouraged to enjoy ice cream, cheesecake, potatoes and mac and cheese, which they can’t do on dialysis because it could lead to a dangerous build-up of potassium and phosphorus. While wearing the WAK, however, the patients maintained equilibrium despite having eaten those foods.
Five of the patients wore the WAK for 24 hours, while two patients stopped wearing the device before the trial ended due to technical issues with the prototype devices. Gura says that he feels confident that those issues will be resolved before the next clinical trial.
“All patients reported that they would switch to the WAK if the device were commercially available,” the authors wrote. If future trials prove successful, it has been given the green-light to be fast-tracked through the U.S. Food and Drug Administration's (FDA) approval process.
How Does It Work?
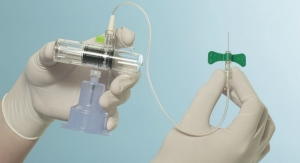
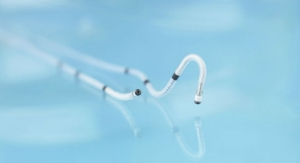
The device connects to a large vein in the body through catheter which has filters to separate water, salts and minerals out of the blood. The filters need to be replaced once a week and chemicals added once per day to allow it to continue filtering the water. The device runs on 9-volt batteries but may be upgraded to run on a rechargeable battery.
The FDA has awarded its new Expedited Access Pathway (EAP) to the externally worn WAK. The EAP designation signals FDA support for expediting the availability of the device to the 600,000+ patients suffering from End-Stage Renal Disease in the United States alone.
“This successful U.S. trial brings us a lot closer to making the WAK available to the public,” said Gura. “Soon, we hope to be improving the lives of hemodialysis patients in the U.S. and around the world with the WAK.”
Nephrologist and inventor of the WAK Victor Gura M.D., who is affiliated with both UCLA and Cedars Sinai Medical Center, presented the results Saturday at the American Society of Nephrology’s annual ASN Kidney Week conference in San Diego, California. The WAK represents the first material advancement in dialysis technology in decades offering new hope for the millions of hemodialysis patients around the globe.
The WAK, which stands for Wearable Artificial Kidney, is a belt-like device that miniaturizes the functions of a 300-pound dialysis machine down to 10 pounds, and the WAK research team believes they can reduce its weight to half that.
It uses just a half liter of water as opposed to 40 gallons per session, and is battery-operated, user-friendly and lightweight allowing for freedom of movement for patients. Much like a normal kidney, the WAK removes excess fluids and toxins from the blood continuously at a natural rate, allowing them to achieve a quality of life closer to that of a healthy person.
Traditional dialysis makes a normal life impossible for patients not only because they need to be strapped to a kidney dialysis machine for 9-12 hours per week but because it severely restricts their diets. With the WAK, patients can enjoy most of the foods forbidden to them for years; improve their quality of life; reduce hospitalizations and increase longevity. Currently, the mortality rate for ESRD patients in the U.S. is at an unacceptable 20%.
Seven patients in Seattle, WA who have end-stage kidney disease (ESRD), took part in the study and were fitted with the device. The patients were encouraged to enjoy ice cream, cheesecake, potatoes and mac and cheese, which they can’t do on dialysis because it could lead to a dangerous build-up of potassium and phosphorus. While wearing the WAK, however, the patients maintained equilibrium despite having eaten those foods.
Five of the patients wore the WAK for 24 hours, while two patients stopped wearing the device before the trial ended due to technical issues with the prototype devices. Gura says that he feels confident that those issues will be resolved before the next clinical trial.
“All patients reported that they would switch to the WAK if the device were commercially available,” the authors wrote. If future trials prove successful, it has been given the green-light to be fast-tracked through the U.S. Food and Drug Administration's (FDA) approval process.
How Does It Work?
The device connects to a large vein in the body through catheter which has filters to separate water, salts and minerals out of the blood. The filters need to be replaced once a week and chemicals added once per day to allow it to continue filtering the water. The device runs on 9-volt batteries but may be upgraded to run on a rechargeable battery.
The FDA has awarded its new Expedited Access Pathway (EAP) to the externally worn WAK. The EAP designation signals FDA support for expediting the availability of the device to the 600,000+ patients suffering from End-Stage Renal Disease in the United States alone.
“This successful U.S. trial brings us a lot closer to making the WAK available to the public,” said Gura. “Soon, we hope to be improving the lives of hemodialysis patients in the U.S. and around the world with the WAK.”