11.11.15
Six months after implantation with an investigational leadless intracardiac transcatheter pacing system from Medtronic plc, 96 percent of patients did not have system-related or procedure-related major complications, according to a prespecified interim analysis of a prospective, multicenter study published online in the New England Journal of Medicine. Further, 98.3 percent of the patients had a low and stable pacing capture threshold, and the device was successfully implanted in 99.2 percent of patients.
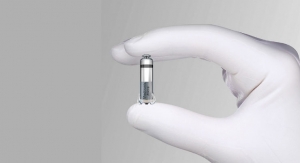
Each of the 725 patients was implanted with the Micra Transcatheter Pacemaker System, a single-chamber ventricular pacemaker that has not yet received U.S. Food and Drug Administration (FDA) approval. Medtronic, the device’s manufacturer, expects to hear about an FDA approval during fiscal year 2017, which begins in May 2016. The Micra received a CE mark in Europe in April.
Medtronic funded the ongoing study, and researchers will follow all of the patients for a year. Medtronic also plans on conducting long-term trials.
Each year, 350,000 patients in the U.S. are implanted with pacemakers, which have been around for more than 50 years.
The researchers enrolled patients at 56 centers in 19 countries who met class I or II guideline-based indications for pacing and were suitable candidates for single-chamber ventricular demand pacing. Patients were excluded if they had a pacemaker or implantable cardioverter-defibrillator. Of the patients, 64 percent had bradycardia associated with persistent or permanent atrial tachyarrhythmia, 17.5 percent had sinus-node dysfunction and 14.8 percent had atrioventricular block.
Clinicians noted there were 28 major complications in 25 patients, including four of the patients who had unsuccessful implantation. The major complications included 11 cardiac injuries, five complications at the groin puncture site, two cases of thromboembolism, two pacing issues and eight other complications. There was one death.
The researchers also mentioned there were no radiographically visible device dislodgements, no telemetry failures and no systemic infections.
In a post hoc analysis, they compared the 725 patients in this study with 2,667 patients who received transvenous pacemakers in six previous trials. The patients who received the Micra transcatheter pacemaker were older and had more coexisting conditions compared with the transvenous pacemaker group.
After six months of follow-up, 4 percent of patients in the Micra transcatheter pacemaker group and 7.4 percent of patients in the transvenous pacemaker group had major complications. In addition, 2.3 percent and 3.9 percent of patients, respectively, were hospitalized and 0.4 percent and 3.5 percent, respectively, had system revisions due to complications.
Each of the 725 patients was implanted with the Micra Transcatheter Pacemaker System, a single-chamber ventricular pacemaker that has not yet received U.S. Food and Drug Administration (FDA) approval. Medtronic, the device’s manufacturer, expects to hear about an FDA approval during fiscal year 2017, which begins in May 2016. The Micra received a CE mark in Europe in April.
Medtronic funded the ongoing study, and researchers will follow all of the patients for a year. Medtronic also plans on conducting long-term trials.
Each year, 350,000 patients in the U.S. are implanted with pacemakers, which have been around for more than 50 years.
The researchers enrolled patients at 56 centers in 19 countries who met class I or II guideline-based indications for pacing and were suitable candidates for single-chamber ventricular demand pacing. Patients were excluded if they had a pacemaker or implantable cardioverter-defibrillator. Of the patients, 64 percent had bradycardia associated with persistent or permanent atrial tachyarrhythmia, 17.5 percent had sinus-node dysfunction and 14.8 percent had atrioventricular block.
Clinicians noted there were 28 major complications in 25 patients, including four of the patients who had unsuccessful implantation. The major complications included 11 cardiac injuries, five complications at the groin puncture site, two cases of thromboembolism, two pacing issues and eight other complications. There was one death.
The researchers also mentioned there were no radiographically visible device dislodgements, no telemetry failures and no systemic infections.
In a post hoc analysis, they compared the 725 patients in this study with 2,667 patients who received transvenous pacemakers in six previous trials. The patients who received the Micra transcatheter pacemaker were older and had more coexisting conditions compared with the transvenous pacemaker group.
After six months of follow-up, 4 percent of patients in the Micra transcatheter pacemaker group and 7.4 percent of patients in the transvenous pacemaker group had major complications. In addition, 2.3 percent and 3.9 percent of patients, respectively, were hospitalized and 0.4 percent and 3.5 percent, respectively, had system revisions due to complications.