10.07.15
Rivanna Medical, a commercial-stage medical device company that has developed a platform of technologies based on medical ultrasound, has rounded out its clinical advisory board with the appointment of Rebecca D. Minehart, M.D., as an advisor. The company's clinical advisory board also includes Brendan Carvalho, M.D., an associate professor at Stanford University's School of Medicine in California, and Thomas R. Hill, M.D., with Newland, N.C.-based Hemlock Medical PLLC.
Minehart is assistant program director in the Department of Anesthesia, Critical Care and Pain Medicine (DACCPM) Anesthesia Residency Program, as well as program director of the Obstetric Anesthesia Fellowship at Massachusetts General Hospital in Boston, and has been a faculty member since August 2007.
“Given that Rivanna's initial commercial application of its Accuro platform, based on automated 3-D navigation, is designed for guiding clinicians to a first-attempt success in administering spinal anesthesia, we believe that Dr. Minehart epitomizes the perfect union of clinical experience and academic prowess to expand our clinical advisory board,” said John A. Williams, Rivanna president and CEO.
According to Williams, the Accuro platform has an addressable $1.1 billion U.S. market. “In light of the significant unmet clinical need for automated image guidance in general, and the spinal anesthesia market in particular, we look forward to soon being able to fast-forward the U.S. commercialization of Accuro, especially with a strategic partner.”
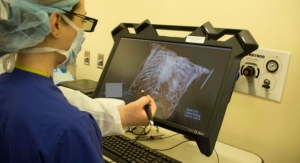
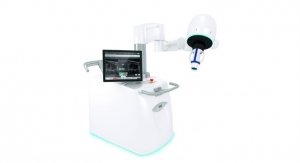
The Accuro device is a pocket-sized, self-contained, battery-operated ultrasound instrument consisting of an ultrasound system, ultrasound probe and rotatable touchscreen display. The products initial application facilitates spinal anesthesia needle guidance with real-time 3-D navigation of the lumbar spine. The technology, based on handheld image automation, is designed to guide a clinician using a needle or probe to a target within the human anatomy. Rivanna executives said the Accuro platform will be leveraged in future products to be used for vascular access, joint injection, thyroid biopsy and nerve blockage.
Spinal anesthesia often fails due to reliance on “blind” needle guidance. There are more than 20 million neuraxial anesthesia-related (epidurals and spinals) procedures per year in the United States; twenty percent to 80 percent of all needle placement attempts fail, depending on operator skill, age, and patient obesity; obese and elderly are growing demographics.
Based in Charlottesville, Va., Rivanna is a privately held medical technology company commercializing its initial product, Accuro, an imaging product that is U.S. Food and Drug Administration 510(k)-cleared for spinal anesthesia guidance and a various additional imaging applications, such as diagnostic imaging of the cardiac and abdominal anatomies.
Minehart is assistant program director in the Department of Anesthesia, Critical Care and Pain Medicine (DACCPM) Anesthesia Residency Program, as well as program director of the Obstetric Anesthesia Fellowship at Massachusetts General Hospital in Boston, and has been a faculty member since August 2007.
“Given that Rivanna's initial commercial application of its Accuro platform, based on automated 3-D navigation, is designed for guiding clinicians to a first-attempt success in administering spinal anesthesia, we believe that Dr. Minehart epitomizes the perfect union of clinical experience and academic prowess to expand our clinical advisory board,” said John A. Williams, Rivanna president and CEO.
According to Williams, the Accuro platform has an addressable $1.1 billion U.S. market. “In light of the significant unmet clinical need for automated image guidance in general, and the spinal anesthesia market in particular, we look forward to soon being able to fast-forward the U.S. commercialization of Accuro, especially with a strategic partner.”
The Accuro device is a pocket-sized, self-contained, battery-operated ultrasound instrument consisting of an ultrasound system, ultrasound probe and rotatable touchscreen display. The products initial application facilitates spinal anesthesia needle guidance with real-time 3-D navigation of the lumbar spine. The technology, based on handheld image automation, is designed to guide a clinician using a needle or probe to a target within the human anatomy. Rivanna executives said the Accuro platform will be leveraged in future products to be used for vascular access, joint injection, thyroid biopsy and nerve blockage.
Spinal anesthesia often fails due to reliance on “blind” needle guidance. There are more than 20 million neuraxial anesthesia-related (epidurals and spinals) procedures per year in the United States; twenty percent to 80 percent of all needle placement attempts fail, depending on operator skill, age, and patient obesity; obese and elderly are growing demographics.
Based in Charlottesville, Va., Rivanna is a privately held medical technology company commercializing its initial product, Accuro, an imaging product that is U.S. Food and Drug Administration 510(k)-cleared for spinal anesthesia guidance and a various additional imaging applications, such as diagnostic imaging of the cardiac and abdominal anatomies.