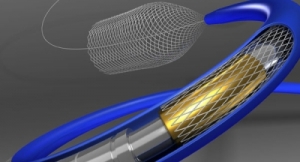
Medtronic plc continues to grow.
The world's largest medical device manufacturer is acquiring private-held Lazarus Effect, developer of a mesh cover for stent retrievers of blood clots, for $100 million in cash. As part of the deal, Medtronic obtains the Lazarus Cover device, which will serve as an adjunct to Medtronic's Solitaire stent retriever device, used to remove clots from acute ischemic stroke patients.
A news release describes the Lazarus Cover as a "novel nitinol ‘mesh cover’ that folds over a stent retriever device during clot retrieval and ‘candy wraps’ the stent with the clot inside." It is designed to trap extra thrombi not captured by the stenting device in the clot retrieval process, thereby improving outcomes of this interventional procedure. The product received CE Mark approval last year, and U.S. Food and Drug Administration (FDA) approval is pending.
Of the 695,000 acute ischemic stroke victims in the United States, about 240,000 are eligible for treatment with a stent retriever, such as Medtronic's Solitaire device, according to the company. About 13,000 procedures using stent retrieval devices were perfomed last year, but that number likely will increase following recent recommendations by the American Heart Association and the American Stroke Association to use such devices as endovascular treatment for acute ischemic stroke in select patients, in addition to standard tissue plasminogen activator (tPA) therapy.
The decision was based on the results of numerous global trials, which showed the use of a stent retrieval device benefits patients who continue to have a blood vessel blocked by a clot even after tPA is administered. At least five of those trials used Medtronic's Solitaire device.
"Medtronic has been a supporter of the recent clinical work showing improved outcomes of ischemic stroke patients treated with endovascular therapy. Their support of data-driven clinical evidence and the success with the Solitaire stent retriever device make them the leader for treating ischemic stroke. Lazarus Effect is pleased to bring our technologies together with Medtronic's therapies," said Martin Dieck, co-founder, president and CEO of Lazarus Effect. "We look forward to working closely with Medtronic to bring this platform forward to the benefit of future patients."
Medtronic executives said the company will fold Lazarus Effect's product line into its Neurovascular division, which is part of the company's Restorative Therapies Group.
"With this acquisition, Medtronic reinforces its commitment to providing solutions to clinicians and patients fighting stroke," said Brett Wall, president of the Neurovascular division at Medtronic. "Lazarus Effect's 'mesh cover' technology complements our ischemic stroke portfolio, and further enhances our Neurovascular business's ability to deliver next-generation technologies."
Since merging with Covidien plc in January and freeing up billions of dollars in overseas cash to spend on acquisitions, Ireland-based Medtronic has been busy snapping up smaller, U.S.-based companies with promising novel device technologies. The purchase of Lazarus Effect is Medtronic's eighth of the year, according to the Star Tribune, which noted the device giant has spent at least $688 million on acquisitions since June.