High-intensity focused ultrasound (HIFU) developer SonaCare Medical is partnering with magnetic resonance imaging (MRI) technology developer Invivo Corporation to use the firm's MRI fusion software with its Sonablate 500 prostate ablation system.
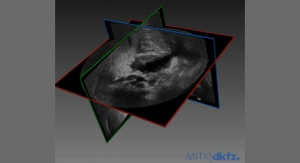
Invivo's software will fuse a preoperative MRI dataset with an intraoperative ultrasound dataset produced by SonaCare's transrectal ablation system, according to SonaCare. The image fusion expands Sonablate users' options for using magnetic resonance (MR) guidance to target areas in the prostate for ablation, the company said.
"Fusing MR images with live ultrasound images allows intraoperative identification and targeting of regions of the prostate not ordinarily seen under operative ultrasound," said SonaCare CEO Mark Carol, M.D. "This process of image fusion provides an improved ability for urologists to plan and target more precisely specific regions of prostate tissue. Adding fusion technology to the ablative capabilities of Sonablate makes it easier for tissue-preserving options, such as focal therapy, to be applied to the prostate. We are pleased to have gained a fusion strategic partner in Invivo in order to enhance further the treatment experience for physicians and patients."
Studies have shown multi-parametric MRI to be an accurate method for identifying regions of abnormal prostate tissue. An annotated and segmented preoperative multi-parametric MRI dataset can be used to guide a targeted biopsy by fusing the MRI to an intraoperative ultrasound dataset generated at the time of biopsy. Adding this same fusion functionally to Sonablate allows an ablation to be targeted in a similar manner, a step considered by many urologists to be critical for the delivery of minimally invasive, tissue preserving, or focal, prostate ablations.
"We look forward to providing Sonablate customers with the best technology available for image visualization and analysis," said Thomas Tynes, senior director of Strategy & Business Development at Invivo. "Today, several hundred healthcare facilities around the world are using Invivo's integrated portfolio of prostate oncology solutions to better visualize and diagnose prostate cancer early. This includes our DynaCAD advanced prostate visualization and analysis software along with our most recent addition - the UroNav Fusion Biopsy System. With the growing adoption of Invivo's prostate oncology solutions and increasing interest in prostate focal therapy, we feel that our mutual customers would benefit greatly from the use of our MRI/US fusion software with the Sonablate 500. The addition of Invivo's fusion software is a natural next step to creating a truly integrated, MRI/US solution to better support and guide the treatment of prostate cancer."
Sonablate 500 has CE marking and is, or has been, approved for use to treat prostate cancer in more than 49 countries outside the United States and is pending de novo submission review by the U.S. Food and Drug Administration (FDA).
SonaCare Medical, a privately held, venture-backed developer of minimally invasive, HIFU technologies. With its subsidiary Focus Surgery Inc., the company designs and manufactures high-intensity focused ultrasound (HIFU) medical devices, including the Sonablate 450, which is investigational in the United States and being studied in a pivotal FDA clinical trial as a possible treatment for recurrent prostate cancer in patients treated previously with external beam radiation therapy; Sonablate 500; and Sonatherm laparoscopic HIFU surgical ablation system, which received FDA 510(k) cleared in the United States, has CE marking and is approved in more than 30 countries worldwide.
SonaCare Medical was founded in 2004 and is headquartered in Charlotte, N.C.