09.03.15
Wayne, Pa.-based Teleflex Inc., which makes medical technologies for critical care and surgery, has received U.S. Food and Drug Administration 510(k) clearance to market its Arrow triple lumen pressure injectable acute hemodialysis catheter.
“To support our goal of providing the right line for the right patient at the right time, adding the Arrow triple lumen pressure injectable acute hemodialysis catheter to our hemodialysis portfolio provides options to clinicians when choosing the best catheter for their patient,” said Jay White, president of the Vascular Division of Teleflex.
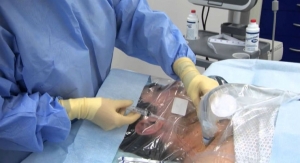
The Arrow triple lumen pressure injectable acute hemodialysis catheter is available in the Arrow Ergopack system, which is meant to help hospitals maintain compliance with current vascular access guidelines and standards while providing maximal barrier protection against infections when inserting these lines.
“To support our goal of providing the right line for the right patient at the right time, adding the Arrow triple lumen pressure injectable acute hemodialysis catheter to our hemodialysis portfolio provides options to clinicians when choosing the best catheter for their patient,” said Jay White, president of the Vascular Division of Teleflex.
The Arrow triple lumen pressure injectable acute hemodialysis catheter is available in the Arrow Ergopack system, which is meant to help hospitals maintain compliance with current vascular access guidelines and standards while providing maximal barrier protection against infections when inserting these lines.