08.05.15
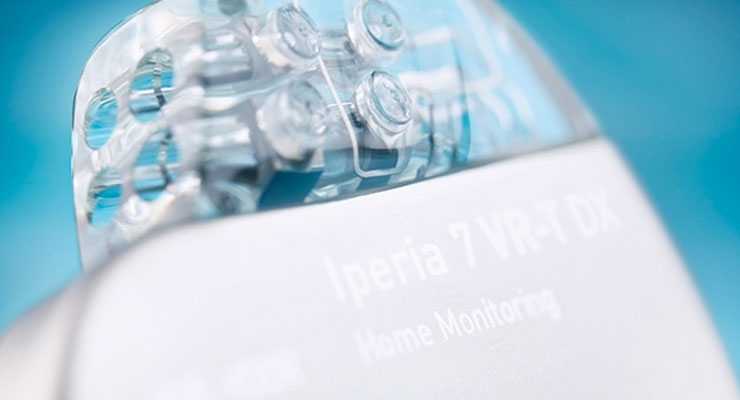
Biotronik SE & Co. KG has launched its Iperia 7 VR-T DX ProMRI single-chamber implantable cardiac defibrillator (ICD) in Japan.
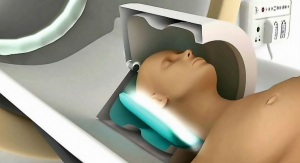
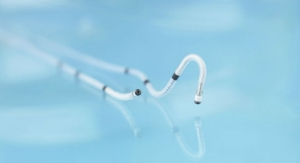
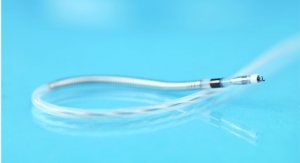
With its DX technology, Biotronik claims its ICDs collect full atrial diagnostic information with just one lead. Executives contends the firm is the first to grant patient access to both ultra-high field 3.0 tesla (T), with an exclusion zone, and full-body magnetic resonance imaging (MRI) at standard 1.5 T scan strength.
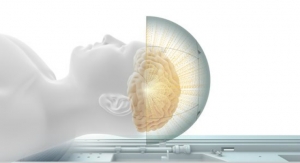
Atrial diagnostic capabilities with a single lead mean there is less likelihood of complications bought about by additional hardware, bigwigs noted in a news release. Allowing patients to undergo 3.0 T MRI scans can be critical in diagnosing conditions such as stroke, brain tumors or orthopedic conditions. Japan is the world's largest MRI market with more MRI scanners per capita than any other country: approximately 47 registered machines per one million people, according to industry statistics.
"Approximately 60 percent of our MR conditional device patients undergo MRI examinations in order to identify potential complications," commented Toshiaki Sato, M.D., from Kyorin University Hospital, Tokyo, Japan, who was one of the first doctors to implant the Iperia DX device in a patient in Japan. "Eighty percent of these scans are brain MRIs. The 3.0 T MRI machine, with improved signal-to-noise ratio, was introduced in our hospital in 2010 and has been used mainly for brain MRI scans. In Japan, 3.0 T compatibility is preferred for MR conditional devices and the need for this technology will be further increased in the future."
With Iperia, inappropriate shocks may be reduced with MorphMatch morphology detection criteria and optimized anti-tachycardia pacing. This makes it easier to give a patient the appropriate level of pacing therapy he or she might need. The device also features Biotronik Home Monitoring, which allows physicians to remotely follow their patients' device status on a daily basis. The system has demonstrated the ability to detect clinically relevant events, including silent, asymptomatic arrhythmias and device related issues, allowing for earlier intervention.
"The DX technology used in Iperia is based on more than a decade of engineering refinement to ensure accurate and reliable sensing of the atrial signal," added Wolf Ruhnke, vice president at Biotronik. "Integrating this with our Home Monitoring System for early detection, and adding the capability of high resolution 3.0 T MRI procedures, now offers patients most complete care and gives physicians the best possible treatment option."
Biotronik's ProMRI technology enables patients with a pacemaker, implantable defibrillator, or cardiac resynchronization therapy defibrillator (CRT-D) or pacemaker (CRT-P) to undergo an MRI scan. ProMRI technology enables pacemaker patients, and now ICD patients, to undergo full-body scans. Patients implanted with an Iperia DX device now also may undergo ultra-high field 3.0 T scans.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.
With its DX technology, Biotronik claims its ICDs collect full atrial diagnostic information with just one lead. Executives contends the firm is the first to grant patient access to both ultra-high field 3.0 tesla (T), with an exclusion zone, and full-body magnetic resonance imaging (MRI) at standard 1.5 T scan strength.
Atrial diagnostic capabilities with a single lead mean there is less likelihood of complications bought about by additional hardware, bigwigs noted in a news release. Allowing patients to undergo 3.0 T MRI scans can be critical in diagnosing conditions such as stroke, brain tumors or orthopedic conditions. Japan is the world's largest MRI market with more MRI scanners per capita than any other country: approximately 47 registered machines per one million people, according to industry statistics.
"Approximately 60 percent of our MR conditional device patients undergo MRI examinations in order to identify potential complications," commented Toshiaki Sato, M.D., from Kyorin University Hospital, Tokyo, Japan, who was one of the first doctors to implant the Iperia DX device in a patient in Japan. "Eighty percent of these scans are brain MRIs. The 3.0 T MRI machine, with improved signal-to-noise ratio, was introduced in our hospital in 2010 and has been used mainly for brain MRI scans. In Japan, 3.0 T compatibility is preferred for MR conditional devices and the need for this technology will be further increased in the future."
With Iperia, inappropriate shocks may be reduced with MorphMatch morphology detection criteria and optimized anti-tachycardia pacing. This makes it easier to give a patient the appropriate level of pacing therapy he or she might need. The device also features Biotronik Home Monitoring, which allows physicians to remotely follow their patients' device status on a daily basis. The system has demonstrated the ability to detect clinically relevant events, including silent, asymptomatic arrhythmias and device related issues, allowing for earlier intervention.
"The DX technology used in Iperia is based on more than a decade of engineering refinement to ensure accurate and reliable sensing of the atrial signal," added Wolf Ruhnke, vice president at Biotronik. "Integrating this with our Home Monitoring System for early detection, and adding the capability of high resolution 3.0 T MRI procedures, now offers patients most complete care and gives physicians the best possible treatment option."
Biotronik's ProMRI technology enables patients with a pacemaker, implantable defibrillator, or cardiac resynchronization therapy defibrillator (CRT-D) or pacemaker (CRT-P) to undergo an MRI scan. ProMRI technology enables pacemaker patients, and now ICD patients, to undergo full-body scans. Patients implanted with an Iperia DX device now also may undergo ultra-high field 3.0 T scans.
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is located in Lake Oswego, Ore.