07.30.15
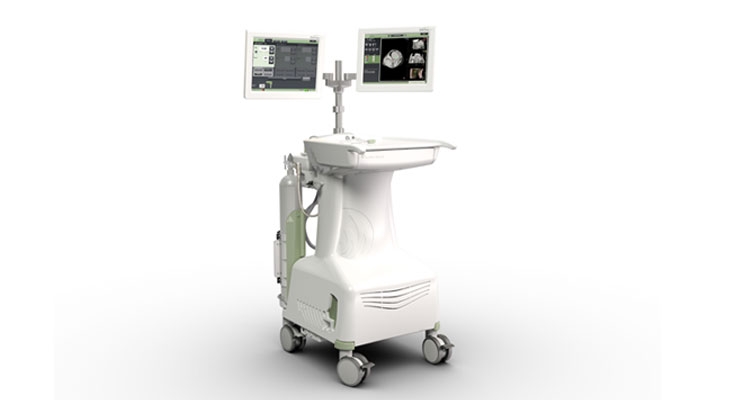
Madison, Wis.-based NeuWave Medical Inc., a privately held medical device company focused on developing ablation technology for soft-tissue lesions has received U.S. Food and Drug Administration (FDA) clearance for its new Ablation Confirmation (AC) software integrated into the NeuWave Intelligent Ablation System.
Ablation Confirmation is a computed tomography (CT) image-processing software package that assists physicians in identifying ablation targets, assessing proper ablation probe placement and confirming ablation zones post-procedure. AC imports images from CT scanners and facility picture archiving and communication systems (PACS) for display and processing during ablation procedures and is accessible to physicians via a dedicated monitor with its own user interface on the NeuWave Intelligent Ablation System. According to company officials, the clearance marks the “first and only, integrated in-procedure ablation confirmation software available on an ablation system.”
“This is a significant step forward, as today physicians performing an ablation have to view patient CT scans with the naked eye on side by side monitors outside the procedure room. Comparing pre- and post-ablation CT scans is cumbersome and requires the user to 'imagine' what the scans would look like if they were overlaid to determine whether or not the lesion has been ablated,” said Dan Sullivan, CEO NeuWave Medical. “Now, with Ablation Confirmation, only available from NeuWave, there is no need to imagine what the ablation scans look like. The system will overlay pre- and post-ablation scans to show the physician whether or not the ablation is complete, all while never leaving the procedure room. This is game changing for physicians and expected to set a new standard of care for the ablation of soft tissue lesions.”
The company also is leveraging its core microwave ablation platform for use with a flexible ablation catheter that is in final stages of development. The initial focus will be on transbronchial, which reflects the approximate 1.8 million new lung lesions diagnosed annually worldwide. Other disease states are expected to follow.
“The vision to integrate Ablation Confirmation software into the ablation system goes back to the company’s inception as we set out to create a system that would set a standard of care. The software is just the first step by NeuWave in developing technology that gives users greater confidence to know if they got it all,” said Dr. Fred Lee, company co-founder and professor of radiology at the University of Wisconsin.
Ablation Confirmation offers:
• In-procedure confirmation: Ablation Confirmation sits resident on the NeuWave Intelligent Ablation System accessing the Hospitals PACS network and the CT scanner in use. AC pulls CT images directly into the software for in-procedure review allowing the physician to review probe placement and ablation confirmation and never break scrub;
• Semi-automated lesion identification: On-screen system drawing tools that can be used to segment the target lesions, add the desired margin and segment the ablation zone.
• 2-D and 3-D visualization: Construction of a two-dimensional and three-dimensional target lesion, probe placement and ablation zone overlay on target.
• Remote collaboration: Remote viewing and collaboration from an external computer without interruption to workflow in the ablation setting.
• Storage of images and procedure data: The system gives the user the option to create snapshots of the entire work flow for transfer back to PACS to use in case reporting, referral follow up, data publication and to establish potential treatment protocols.
The NeuWave Medical Intelligent Ablation System currently is used by physicians in more than half of the top cancer institutions in the United States to ablate a variety of benign and malignant soft-tissue lesions, the company claims. NeuWave’s microwave ablation platform works by delivering focused heat to eliminate or “ablate” lesions in the body. One or more antennas, often called “probes,” are placed directly into the lesion and then emit microwave energy from the tip, rapidly oscillating water molecules within the lesion. This oscillation causes friction and enough heat to destroy the lesion. A versatile probe portfolio allows the treating physician to tailor the approach to each patient, maximizing energy delivery and ablating the lesion, while helping to protect non-targeted tissue in the body.
Microwave ablation is most commonly performed as an outpatient procedure done under general anesthesia or moderate/conscious sedation and is covered under a category 1 CPT code. A widely adopted and accepted treatment modality, the National Comprehensive Cancer Network, which authors the most practiced guidelines, supports the use of percutaneous thermal ablation for lung, kidney and liver lesions.
The company also has an office in Minneapolis, Minn.
Ablation Confirmation is a computed tomography (CT) image-processing software package that assists physicians in identifying ablation targets, assessing proper ablation probe placement and confirming ablation zones post-procedure. AC imports images from CT scanners and facility picture archiving and communication systems (PACS) for display and processing during ablation procedures and is accessible to physicians via a dedicated monitor with its own user interface on the NeuWave Intelligent Ablation System. According to company officials, the clearance marks the “first and only, integrated in-procedure ablation confirmation software available on an ablation system.”
“This is a significant step forward, as today physicians performing an ablation have to view patient CT scans with the naked eye on side by side monitors outside the procedure room. Comparing pre- and post-ablation CT scans is cumbersome and requires the user to 'imagine' what the scans would look like if they were overlaid to determine whether or not the lesion has been ablated,” said Dan Sullivan, CEO NeuWave Medical. “Now, with Ablation Confirmation, only available from NeuWave, there is no need to imagine what the ablation scans look like. The system will overlay pre- and post-ablation scans to show the physician whether or not the ablation is complete, all while never leaving the procedure room. This is game changing for physicians and expected to set a new standard of care for the ablation of soft tissue lesions.”
The company also is leveraging its core microwave ablation platform for use with a flexible ablation catheter that is in final stages of development. The initial focus will be on transbronchial, which reflects the approximate 1.8 million new lung lesions diagnosed annually worldwide. Other disease states are expected to follow.
“The vision to integrate Ablation Confirmation software into the ablation system goes back to the company’s inception as we set out to create a system that would set a standard of care. The software is just the first step by NeuWave in developing technology that gives users greater confidence to know if they got it all,” said Dr. Fred Lee, company co-founder and professor of radiology at the University of Wisconsin.
Ablation Confirmation offers:
• In-procedure confirmation: Ablation Confirmation sits resident on the NeuWave Intelligent Ablation System accessing the Hospitals PACS network and the CT scanner in use. AC pulls CT images directly into the software for in-procedure review allowing the physician to review probe placement and ablation confirmation and never break scrub;
• Semi-automated lesion identification: On-screen system drawing tools that can be used to segment the target lesions, add the desired margin and segment the ablation zone.
• 2-D and 3-D visualization: Construction of a two-dimensional and three-dimensional target lesion, probe placement and ablation zone overlay on target.
• Remote collaboration: Remote viewing and collaboration from an external computer without interruption to workflow in the ablation setting.
• Storage of images and procedure data: The system gives the user the option to create snapshots of the entire work flow for transfer back to PACS to use in case reporting, referral follow up, data publication and to establish potential treatment protocols.
The NeuWave Medical Intelligent Ablation System currently is used by physicians in more than half of the top cancer institutions in the United States to ablate a variety of benign and malignant soft-tissue lesions, the company claims. NeuWave’s microwave ablation platform works by delivering focused heat to eliminate or “ablate” lesions in the body. One or more antennas, often called “probes,” are placed directly into the lesion and then emit microwave energy from the tip, rapidly oscillating water molecules within the lesion. This oscillation causes friction and enough heat to destroy the lesion. A versatile probe portfolio allows the treating physician to tailor the approach to each patient, maximizing energy delivery and ablating the lesion, while helping to protect non-targeted tissue in the body.
Microwave ablation is most commonly performed as an outpatient procedure done under general anesthesia or moderate/conscious sedation and is covered under a category 1 CPT code. A widely adopted and accepted treatment modality, the National Comprehensive Cancer Network, which authors the most practiced guidelines, supports the use of percutaneous thermal ablation for lung, kidney and liver lesions.
The company also has an office in Minneapolis, Minn.