07.14.15
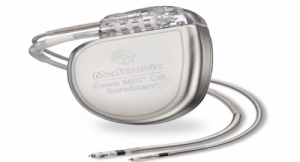
Biotronik SE & Co. KG said the Food and Drug Administration (FDA) has approved its implantable cardioverter defibrillator (ICD) that can deliver ultra-high energy on the first shock. Also included in the approval response from the FDA is the latest generation of defibrillator devices for patients with complex heart rhythm conditions.
Two additional options of this defibrillator hybrid, the next generation Biotronik DX systems, are now available to U.S. patients. They are:
DX technology is based on more than a decade of engineering refinement to ensure accurate and reliable sensing and processing of the atrial signal, according to the company. Due to its unique technology, the DX System has continued to drive Biotronik’s U.S. customer adoption since it initially was introduced in February 2013.
It is the first defibrillator system equipped to provide dual-chamber diagnostic information with just one specialized defibrillator lead, Biotronik officials claim. Physicians consider the DX System a solution to the limited capabilities of single chamber devices, and several potential complications associated with dual chamber devices.
“Physician experts in the field of electrophysiology have indicated that meaningful therapeutic innovations available in Itrevia and Inventra ICDs - like high energy options, DX systems and CLS - make it easier for physicians to tailor therapy and improve the quality of life for their patients,” said Marlou Janssen, president of Biotronik. “This approval is an example of Biotronik's ability to offer stand-alone technology to support electrophysiologists as they pursue their work for patients.”
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is in Lake Oswego, Ore.
Two additional options of this defibrillator hybrid, the next generation Biotronik DX systems, are now available to U.S. patients. They are:
- Inventra DX – the only such ICD system that can deliver ultra-high energy on the first shock; offering better protection to patients that depend on this life-saving therapy.
-
Itrevia DX – a less invasive therapy with an algorithm that can identify successful pain-free therapies for each patient and dynamically re-apply them as needed. The complete Itrevia family - including Itrevia VR-T, Itrevia DR-T, and Itrevia HF-T – were approved together.
DX technology is based on more than a decade of engineering refinement to ensure accurate and reliable sensing and processing of the atrial signal, according to the company. Due to its unique technology, the DX System has continued to drive Biotronik’s U.S. customer adoption since it initially was introduced in February 2013.
It is the first defibrillator system equipped to provide dual-chamber diagnostic information with just one specialized defibrillator lead, Biotronik officials claim. Physicians consider the DX System a solution to the limited capabilities of single chamber devices, and several potential complications associated with dual chamber devices.
“Physician experts in the field of electrophysiology have indicated that meaningful therapeutic innovations available in Itrevia and Inventra ICDs - like high energy options, DX systems and CLS - make it easier for physicians to tailor therapy and improve the quality of life for their patients,” said Marlou Janssen, president of Biotronik. “This approval is an example of Biotronik's ability to offer stand-alone technology to support electrophysiologists as they pursue their work for patients.”
Biotronik is based in Berlin, Germany, and has a global workforce of more than 5,600 employees. Its U.S. headquarters is in Lake Oswego, Ore.