07.08.15
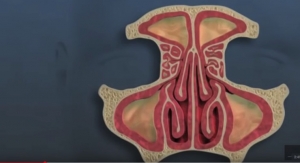
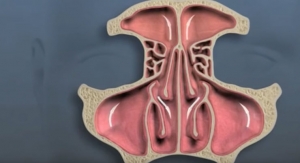
Menlo Park, Calif.-based Intersect ENT Inc., which makes technology for the ear, nose and throat (ENT) market, has enrolled the first patient in the pivotal study of its latest bioabsorbable steroid releasing sinus implant, currently called Nova. Nova is designed to mechanically prop sinuses open while delivering anti-inflammatory medication following surgical interventions such as sinus surgery in the operating room or in-office sinus dilation. Nova has an hourglass shape and lower profile, meant to allow for placement in the smaller frontal and maxillary sinus openings, and which may expand the applicable patient population for steroid releasing implants.
The Nova study is a prospective, randomized, blinded, multi-center trial of 80 patients designed to assess implant safety and efficacy. The company is conducting the Nova trial as a second cohort of patients of the Progress study.
The first cohort of the Progress study was an 80-patient prospective, randomized blinded multi-center trial to assess the safety and efficacy of the Propel mini drug eluting implant to improve outcomes following frontal sinus surgery and was designed to support an expanded indication for Propel mini, currently approved for use in the ethmoid sinus.
In addition to the beginning of patient enrollment in the Nova study, the company also announced that enrollment in the Propel mini cohort of the study has recently been completed.
“Clinical evidence to date has shown that Propel and Propel mini meaningfully improve the outcomes of patients undergoing ethmoid sinus surgery. Patients with chronic sinusitis may benefit similarly from a steroid-releasing implant as part of treatment of the frontal sinus,” said Tim Smith, M.D., M.P.H., F.A.C.S, a cancer and ENT specialist at Oregon Health and Science University, who serves as the principal investigator of the study. “I am pleased to be involved with the clinical assessment of both Propel mini and Nova and believe that each of these products has the potential to offer a differentiated solution in the continuum of care for patients with chronic sinusitis.”
“We are excited to commence enrollment in our pivotal study of Nova and to have completed enrollment in the Propel mini cohort of the Progress trial,” said Lisa Earnhardt, president and CEO of Intersect ENT. “Intersect ENT is committed to furthering clinical evidence that expands options for people suffering from sinusitis, a chronic condition that severely impacts quality of life.”
The Nova study is a prospective, randomized, blinded, multi-center trial of 80 patients designed to assess implant safety and efficacy. The company is conducting the Nova trial as a second cohort of patients of the Progress study.
The first cohort of the Progress study was an 80-patient prospective, randomized blinded multi-center trial to assess the safety and efficacy of the Propel mini drug eluting implant to improve outcomes following frontal sinus surgery and was designed to support an expanded indication for Propel mini, currently approved for use in the ethmoid sinus.
In addition to the beginning of patient enrollment in the Nova study, the company also announced that enrollment in the Propel mini cohort of the study has recently been completed.
“Clinical evidence to date has shown that Propel and Propel mini meaningfully improve the outcomes of patients undergoing ethmoid sinus surgery. Patients with chronic sinusitis may benefit similarly from a steroid-releasing implant as part of treatment of the frontal sinus,” said Tim Smith, M.D., M.P.H., F.A.C.S, a cancer and ENT specialist at Oregon Health and Science University, who serves as the principal investigator of the study. “I am pleased to be involved with the clinical assessment of both Propel mini and Nova and believe that each of these products has the potential to offer a differentiated solution in the continuum of care for patients with chronic sinusitis.”
“We are excited to commence enrollment in our pivotal study of Nova and to have completed enrollment in the Propel mini cohort of the Progress trial,” said Lisa Earnhardt, president and CEO of Intersect ENT. “Intersect ENT is committed to furthering clinical evidence that expands options for people suffering from sinusitis, a chronic condition that severely impacts quality of life.”