07.06.15
Direct Flow Medical Inc. has received Investigational Device Exemption approval from the U.S. Food and Drug Administration (FDA) to broaden its SALUS Trial, including the addition of high risk patients and randomization against a commercial device, the Medtronic CoreValve.
The SALUS Trial is a prospective, randomized, multi-center, core lab adjudicated U.S. clinical trial, evaluating the Direct Flow Medical Transcatheter Aortic Valve System. The primary endpoint of the trial is a composite of all-cause mortality and disabling stroke at 12 months. Principal investigators for the SALUS Trial are Murat Tuzcu, M.D., vice chairman of the Department of Cardiology at the Cleveland Clinic, and Patrick McCarthy, M.D., director of the Bluhm Cardiovascular Institute and chief of Cardiac Surgery, Northwestern Memorial Hospital in Chicago, Ill.
The expansion of the SALUS Trial includes the following changes:
“We have been working closely with the FDA since the early development of the Direct Flow Medical technology and it has been a collaborative effort with the FDA which has led to an expansion of the SALUS trial. The technology has shown outstanding performance in clinical trials and commercial settings, reducing the risk of aortic regurgitation with survival. We look forward to expanding the clinical use to additional centers in the U.S.,” said Tuzcu.
An earlier feasibility phase of the SALUS Trial conducted in 2013 evaluated the system in 30 patients. The 30-day outcomes demonstrated a survival rate of 97 percent with 100 percent of patients experiencing mild or less aortic regurgitation. Results also showed low procedural complications, no incidence of stroke and a 3 percent rate of permanent pacing. The mean aortic gradient decreased from 44.5 mmHg to 12.7 mmHg at 30 days. The previous protocol of the SALUS Trial has enrolled more than 100 extreme-risk patients in a non-randomized fashion and will continued to be followed independently.
The Direct Flow Medical system received the CE Mark in January 2013 and currently is available commercially in Europe.
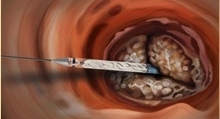
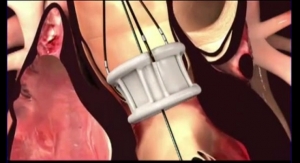
The Direct Flow Medical Transcatheter Aortic Valve System features a metal-free frame. Rather than a metal stent, the Direct Flow Medical System incorporates a polymer frame, which is expanded using pressurized saline and contrast for placement, assessment and repositioning. The saline/contrast solution is exchanged for a polymer that solidifies and secures the valve in place once optimal positioning is reached. The unique double-ring design of the valve creates a tight seal around the annulus, according to the company. The system is fully repositionable and retrievable up until polymer exchange. The metal-free design enables a low-profile (18 French), fully sheathed delivery system for all valve sizes that minimizes vascular complications and improves hemodynamic outcomes.
Founded in 2004, Direct Flow Medical develops transcatheter heart valve technologies. The company is headquartered in Santa Rosa, Calif., with technology and manufacturing facilities in Lake Forest, Calif. Direct Flow Medical investors include EDF Ventures, New Leaf Venture Partners, Spray Venture Partners, Foundation Medical Partners, VantagePoint Venture Partners, ePlanet Venture Partners, SV Life Sciences and strategic corporate investors.
The SALUS Trial is a prospective, randomized, multi-center, core lab adjudicated U.S. clinical trial, evaluating the Direct Flow Medical Transcatheter Aortic Valve System. The primary endpoint of the trial is a composite of all-cause mortality and disabling stroke at 12 months. Principal investigators for the SALUS Trial are Murat Tuzcu, M.D., vice chairman of the Department of Cardiology at the Cleveland Clinic, and Patrick McCarthy, M.D., director of the Bluhm Cardiovascular Institute and chief of Cardiac Surgery, Northwestern Memorial Hospital in Chicago, Ill.
The expansion of the SALUS Trial includes the following changes:
- Treatment of high surgical risk patients and continued treatment of extreme risk patients
- 2:1 randomization to Medtronic CoreValve
- 912-subject pivotal cohort
- Up to 45 sites in the United States
- Registries for non-femoral access points and type 2 and 3 bicuspid valves
“We have been working closely with the FDA since the early development of the Direct Flow Medical technology and it has been a collaborative effort with the FDA which has led to an expansion of the SALUS trial. The technology has shown outstanding performance in clinical trials and commercial settings, reducing the risk of aortic regurgitation with survival. We look forward to expanding the clinical use to additional centers in the U.S.,” said Tuzcu.
An earlier feasibility phase of the SALUS Trial conducted in 2013 evaluated the system in 30 patients. The 30-day outcomes demonstrated a survival rate of 97 percent with 100 percent of patients experiencing mild or less aortic regurgitation. Results also showed low procedural complications, no incidence of stroke and a 3 percent rate of permanent pacing. The mean aortic gradient decreased from 44.5 mmHg to 12.7 mmHg at 30 days. The previous protocol of the SALUS Trial has enrolled more than 100 extreme-risk patients in a non-randomized fashion and will continued to be followed independently.
The Direct Flow Medical system received the CE Mark in January 2013 and currently is available commercially in Europe.
The Direct Flow Medical Transcatheter Aortic Valve System features a metal-free frame. Rather than a metal stent, the Direct Flow Medical System incorporates a polymer frame, which is expanded using pressurized saline and contrast for placement, assessment and repositioning. The saline/contrast solution is exchanged for a polymer that solidifies and secures the valve in place once optimal positioning is reached. The unique double-ring design of the valve creates a tight seal around the annulus, according to the company. The system is fully repositionable and retrievable up until polymer exchange. The metal-free design enables a low-profile (18 French), fully sheathed delivery system for all valve sizes that minimizes vascular complications and improves hemodynamic outcomes.
Founded in 2004, Direct Flow Medical develops transcatheter heart valve technologies. The company is headquartered in Santa Rosa, Calif., with technology and manufacturing facilities in Lake Forest, Calif. Direct Flow Medical investors include EDF Ventures, New Leaf Venture Partners, Spray Venture Partners, Foundation Medical Partners, VantagePoint Venture Partners, ePlanet Venture Partners, SV Life Sciences and strategic corporate investors.