06.15.15
Accurexa Inc.—a biotechnology company focused on developing neurological therapies—has filed a submission with the U.S. Food and Drug Administration (FDA) for 510(k) clearance to market its BranchPoint device—the first in the startup's technology pipeline.
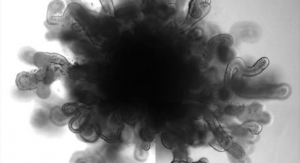
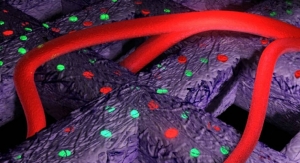
BranchPoint device was invented to deliver therapeutics, such as stem cells, through the radial deployment of a flexible catheter to specific brain target areas through a single brain penetration. The current standard of care is the use of straight, rigid needles, often requiring surgeons to penetrate the brain multiple times for the delivery of therapeutics, which in turn may increase the risk of bleeding, stroke and reflux of therapeutics back out to the brain surface.
A study at University of California, San Francisco (UCSF) demonstrated that the use of a straight needle was associated with reflux of at least 75 percent of the infusion while no reflux was found with the BranchPoint design. Subsequent studies demonstrated that the BranchPoint device can function with interventional MRI guidance, which may further improve the efficiency of delivery.
"We are excited that we reached this important milestone today. We believe that this accomplishment demonstrates that our team at Accurexa is capable of bringing medical innovations to a regulatory submission to the FDA," said George Yu, president and CEO.
"I want to thank my research team, UCSF and everybody who was involved in this endeavor over the last several years. I also want to thank the California Institute of Regenerative Medicine, which provided nearly $2 million in support for the development of the BranchPoint device. If approved by the FDA, it would enable us to leverage this significant research investment for the clinical treatment of a wide range of diseases of the brain," said Daniel Lim, M.D., Ph.D., who is associate professor of neurological surgery at UCSF and the lead inventor of the BranchPoint technology.
The BranchPoint device can deliver therapeutics through the radial deployment of a flexible delivery catheter to large and anatomically complex brain targets through a single initial brain penetration. Clinicians can "tailor" therapeutic delivery to individual patient anatomy and specific disease targets, which may enhance the efficacy of therapies. The company is also developing its ACX-31 program which could deliver temozolomide, a chemotherapy drug, directly to brain cancer sites with its BranchPoint device. Temozolomide is a generic, approved, first-line chemotherapy drug that is indicated for the treatment of adult patients with newly diagnosed glioblastoma multiforme concomitantly with radiotherapy and then as maintenance treatment.
Before temozolomide became generic, it generated U.S. sales of $420 million and global sales of $910 million under its brand name Temodar in 2012. However, current standard of care of delivering temozolomide to tumor sites through oral administration is limited by the blood-brain-barrier and orally administered temozolomide increases patient survival by a significant but modest 2.5 months from a 12.1 months median overall survival.
BranchPoint device was developed at UCSF with $1.8 million in funding from the California Institute for Regenerative Medicine. It is based on a neurosurgical delivery platform that the company exclusively licensed from the University. According to company officials, it can enable new approaches to neurological therapy and be modified for the delivery of a wide range of novel therapeutics, such as chemotherapeutics to brain tumors, stem cells to treat neurodegenerative diseases, or gene therapy.
BranchPoint device was invented to deliver therapeutics, such as stem cells, through the radial deployment of a flexible catheter to specific brain target areas through a single brain penetration. The current standard of care is the use of straight, rigid needles, often requiring surgeons to penetrate the brain multiple times for the delivery of therapeutics, which in turn may increase the risk of bleeding, stroke and reflux of therapeutics back out to the brain surface.
A study at University of California, San Francisco (UCSF) demonstrated that the use of a straight needle was associated with reflux of at least 75 percent of the infusion while no reflux was found with the BranchPoint design. Subsequent studies demonstrated that the BranchPoint device can function with interventional MRI guidance, which may further improve the efficiency of delivery.
"We are excited that we reached this important milestone today. We believe that this accomplishment demonstrates that our team at Accurexa is capable of bringing medical innovations to a regulatory submission to the FDA," said George Yu, president and CEO.
"I want to thank my research team, UCSF and everybody who was involved in this endeavor over the last several years. I also want to thank the California Institute of Regenerative Medicine, which provided nearly $2 million in support for the development of the BranchPoint device. If approved by the FDA, it would enable us to leverage this significant research investment for the clinical treatment of a wide range of diseases of the brain," said Daniel Lim, M.D., Ph.D., who is associate professor of neurological surgery at UCSF and the lead inventor of the BranchPoint technology.
The BranchPoint device can deliver therapeutics through the radial deployment of a flexible delivery catheter to large and anatomically complex brain targets through a single initial brain penetration. Clinicians can "tailor" therapeutic delivery to individual patient anatomy and specific disease targets, which may enhance the efficacy of therapies. The company is also developing its ACX-31 program which could deliver temozolomide, a chemotherapy drug, directly to brain cancer sites with its BranchPoint device. Temozolomide is a generic, approved, first-line chemotherapy drug that is indicated for the treatment of adult patients with newly diagnosed glioblastoma multiforme concomitantly with radiotherapy and then as maintenance treatment.
Before temozolomide became generic, it generated U.S. sales of $420 million and global sales of $910 million under its brand name Temodar in 2012. However, current standard of care of delivering temozolomide to tumor sites through oral administration is limited by the blood-brain-barrier and orally administered temozolomide increases patient survival by a significant but modest 2.5 months from a 12.1 months median overall survival.
BranchPoint device was developed at UCSF with $1.8 million in funding from the California Institute for Regenerative Medicine. It is based on a neurosurgical delivery platform that the company exclusively licensed from the University. According to company officials, it can enable new approaches to neurological therapy and be modified for the delivery of a wide range of novel therapeutics, such as chemotherapeutics to brain tumors, stem cells to treat neurodegenerative diseases, or gene therapy.