06.11.15
Penumbra Inc. has received 510(k) clearance from the U.S. Food and Drug Administration to market its ACE64 aspiration thrombectomy system for the revascularization of large vessel occlusions in patients with acute ischemic stroke.
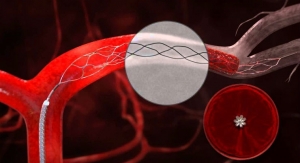
The efficacy and safety of mechanical thrombectomy has been demonstrated in the MR CLEAN trial and confirmed in additional randomized controlled stroke trials, the company said in a news release about the approval. Penumbra bigwigs tout the ACE64 as "the most advanced innovation in thrombectomy technology" since the trials were conducted. Built to leverage breakthrough technology in materials science, ACE64 enables physicians to bring the most powerful clot extraction capability directly to the occlusion and remove clot en masse. Early experience from a European multicenter study, where ACE64 already is available, reported high rates of revascularization at 96 percent TICI 2b/3, a fast procedure time of 37 minutes on average and mRS scores ≤2 at discharge of 48 percent.
“ACE64 is the latest technology improvement in mechanical thrombectomy,” said Rob T. Lo, M.D., of University Medical Center Utrecht in The Netherlands, a center in the MR CLEAN trial. “I have used Merci and the different stent retrievers as well as the prior ACE aspiration thrombectomy system. With the new ACE64, I am achieving even higher revascularization rates, particularly TICI 3, while reducing procedure times and minimizing overall procedure costs. ACE64 is now my frontline tool for treating patients with acute ischemic stroke.”
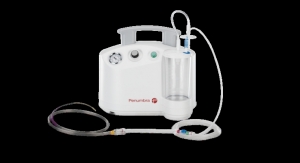
Penumbra’s aspiration thrombectomy devices use a minimally invasive “vacuum” inside the artery to remove a blood clot. The next-generation ACE64 features an even larger aspiration lumen compared with ACE to evacuate large clot burdens, company executives claim.
“We are at an exciting moment in history when the effectiveness of mechanical thrombectomy is now firmly established, allowing the stroke community to focus on optimizing the delivery of care in stroke and improving patient outcomes,” said Adam Elsesser, Penumbra chairman/CEO.
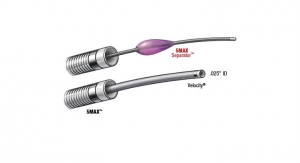
ACE64 features the largest lumen aspiration thrombectomy device on the market with a 0.064-inch distal inner diameter and a 0.068-inch proximal inner diameter, designed to evacuate large clot burdens from the neurovasculature. Built on the ACE tracking technology platform, ACE64 enables optimal clot engagement for even faster and more complete clot removal. ACE64 received CE Mark in December 2014 and is intended for acute ischemic patients with large vessel occlusive disease within eight hours of symptom onset. Aspiration thrombectomy with the ACE64 represents the foundation of intra-arterial therapy for acute ischemic stroke patients, working seamlessly with adjunct devices to simplify procedures and lower procedural costs.
Penumbra designs, develops, manufactures and markets neuro and peripheral vascular devices, targeting (among others) ischemic stroke, hemorrhagic stroke and various peripheral vascular conditions that can be treated through thrombectomy and embolization. The company has more than 850 employees and is based in Alameda, Calif.
The efficacy and safety of mechanical thrombectomy has been demonstrated in the MR CLEAN trial and confirmed in additional randomized controlled stroke trials, the company said in a news release about the approval. Penumbra bigwigs tout the ACE64 as "the most advanced innovation in thrombectomy technology" since the trials were conducted. Built to leverage breakthrough technology in materials science, ACE64 enables physicians to bring the most powerful clot extraction capability directly to the occlusion and remove clot en masse. Early experience from a European multicenter study, where ACE64 already is available, reported high rates of revascularization at 96 percent TICI 2b/3, a fast procedure time of 37 minutes on average and mRS scores ≤2 at discharge of 48 percent.
“ACE64 is the latest technology improvement in mechanical thrombectomy,” said Rob T. Lo, M.D., of University Medical Center Utrecht in The Netherlands, a center in the MR CLEAN trial. “I have used Merci and the different stent retrievers as well as the prior ACE aspiration thrombectomy system. With the new ACE64, I am achieving even higher revascularization rates, particularly TICI 3, while reducing procedure times and minimizing overall procedure costs. ACE64 is now my frontline tool for treating patients with acute ischemic stroke.”
Penumbra’s aspiration thrombectomy devices use a minimally invasive “vacuum” inside the artery to remove a blood clot. The next-generation ACE64 features an even larger aspiration lumen compared with ACE to evacuate large clot burdens, company executives claim.
“We are at an exciting moment in history when the effectiveness of mechanical thrombectomy is now firmly established, allowing the stroke community to focus on optimizing the delivery of care in stroke and improving patient outcomes,” said Adam Elsesser, Penumbra chairman/CEO.
ACE64 features the largest lumen aspiration thrombectomy device on the market with a 0.064-inch distal inner diameter and a 0.068-inch proximal inner diameter, designed to evacuate large clot burdens from the neurovasculature. Built on the ACE tracking technology platform, ACE64 enables optimal clot engagement for even faster and more complete clot removal. ACE64 received CE Mark in December 2014 and is intended for acute ischemic patients with large vessel occlusive disease within eight hours of symptom onset. Aspiration thrombectomy with the ACE64 represents the foundation of intra-arterial therapy for acute ischemic stroke patients, working seamlessly with adjunct devices to simplify procedures and lower procedural costs.
Penumbra designs, develops, manufactures and markets neuro and peripheral vascular devices, targeting (among others) ischemic stroke, hemorrhagic stroke and various peripheral vascular conditions that can be treated through thrombectomy and embolization. The company has more than 850 employees and is based in Alameda, Calif.