Two Watertown, Mass.-based medical device firms, Arsenal Medical and 480 Biomedical, received $26.5 million in combined funding a group of investors.
Arsenal Medical, a company developing novel, polymer based foam and nanofiber products, raised $16 million from Polaris Partners, North Bridge Venture Partners, and Intersouth Partners. 480 Biomedical, a clinical-stage company developing innovative bioresorbable scaffold products, raised $10.5 million from the same syndicate in conjunction with a long-term strategic investor. Both rounds included a conversion of debt, in addition to new equity financing.
The companies, which spun out one from the other in late 2011 and continue to share lab space and resources, are jointly led by President and CEO Maria Palasis, who assumed leadership of both organizations in January.
“Both 480 Biomedical and Arsenal Medical have advanced innovation down exciting yet different paths,” said Palasis. “These organizations have made tremendous strides in their respective programs over the last several years, and the promise of our platforms has never been greater. The commitment from our investors has allowed us to reach our critical milestones across both organizations, and we expect this momentum to continue.”
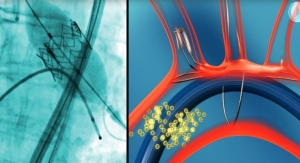
Arsenal Medical is developing two technology platforms: an in-situ forming foam technology and a coaxial nanofiber technology. The in-situ forming foams provide life-saving therapy for acute abdominal hemorrhage and are expected to enter the clinic later this year. Foams also are being developed for other critical clinical conditions including vascular disease and bleeding. The nanofiber technology platform for controlled drug delivery has recently entered into two strategic relationships with major pharmaceutical partners.
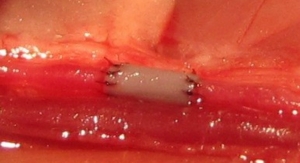
480 Biomedical is advancing the first fully self-expanding, bioresorbable scaffold platform technology to treat vascular disease in adult and pediatric patients. According to the company, the technology provides “the unique ability to support the vessel during healing and leave nothing behind post treatment due to its bioresorbability.” The company’s lead application is the treatment of adult superficial femoral artery disease using a drug-eluting scaffold, and is currently being studied in a global clinical study. For pediatric patients, the company has advanced the development of a bioresorbable scaffold to treat pulmonary artery stenosis in patients who have limited treatment options.
“As the chairman and an investor, it is tremendously gratifying to be a part of two companies that are transforming medical devices through innovations in materials science,” said Carmichael Roberts of North Bridge Venture Partners. “The Arsenal Medical and 480 Biomedical teams have proven themselves to be highly efficient with their resources while pursuing novel breakthrough products that are poised to have significant and positive impacts on large patient populations. We are proud to have supported their efforts from the beginning and look forward to the next phase of their journey.”
Prior to joining the companies, Palasis was research and development director at Boston Scientific Corp., where she managed a portfolio of external biotech and medical device investments and was responsible for the development of new convergence products. She was as an early member of the team that created the Taxus drug-eluting stent.